Abstract
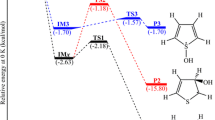
Fukui's IRC is obtained for the pyrolysis of ethyl formate within the level of MNDO molecular orbital calculations. The activation enthalpy and entropy can be evaluated from the calculated partition functions and compared with the observed values. It is suggested that the pyrolysis would be significantly promoted when it occurs in an aprotic polar solvent.
Similar content being viewed by others
References
Fukui, K.: J. Phys. Chem. 74, 4161 (1970).
For instance, see Fukui, K.: “The World of Quantum Chemistry”, Reidel 1974 p. 113; Fukui, K., Kato, S., Fujimoto, H.: J. Am. Chem. Soc. 97, 1 (1975); Kato, S., Fukui, K.: Am. Chem. Soc. 98, 6395 (1976); Kato, S., Kato, H., Fukui, K.: Am. Chem. Soc. 99, 684 (1977); Fukui, K., Koga, N., Fujimoto, H.: ibid. 103, 196 (1981); Tachibana, A., Fukui, K.: Theoret Chim. Acta (Berl.) 49, 321 (1978); ibid. 51, 189 (1979); Theoret. Chim. Acta (Berl.) 51, 275 (1979); ibid., 57, 81 (1980); Yamashita, K., Kaminoyama, M., Yamabe, T., Fukui, K.: Theoret. Chim. Acta (Berl.) 60, 303 (1981); Fujimoto, H., Fukui, K.: Israel J. Chem. 19, 317 (1980); Fujimoto, H., Koga, N., Endo, M., Fukui, K.: Tetrahedron Letts. 22, 1263 (1981)
McIver, J. W., Komornicki, A.: J. Am. Chem. Soc. 94, 2625 (1972)
Ishida, K., Morokuma, K., Komornicki, A.; J. Chem. Phys. 66, 2153 (1977)
Chuchani, G., Martin, I., Hernandez, J. A., Rotinov, A., Fraile, G., Bigley, D. B.: J. Phys. Chem. 84, 944 (1980)
Martin, I., Chuchani, G., Avlla, I., Rotinov, A., Olmos, R.: J. Phys. Chem. 84, 9 (1980)
For instance, see Maccoll, A., Nagra, S. S.: J. Chem. Soc. Faraday trans. I 1975, 2450
Dewar, M. J. S., Thiel, W.: J. Am. Chem. Soc. 99, 4899 (1977); Q. C. P. E. No. 353
Dewar, M. J. S., Ford, G. P., McKee, M. L., Rzepa, H. S., Thiel, W., Yamaguchi, Y.: J. Mol. Struc. 43, 135 (1978)
For instance, see Nagase, S., Ray, N. K., Morokuma, K.: J. Am. Chem. Soc. 102, 4536 (1980)
Herzberg, G.: Molecular spectra and molecular structures. Vol. II. Infrared and raman spectra of polyatomic molecules. Chap. V, London: Van Nostrand Co. Inc., 1945
Maccoll A.: J. Chem. Soc. 1958, 3398
Golden, D. M.: J. Phys. Chem. 83, 108 (1979)
For instance, see Zielinski, T. J., Breen, D. L., Rein, R.: J. Am. Chem. Soc. 100, 6266 (1978)
Klopman, G., Andreozzi, P., Hopfinger, A. J., Kikuchi, O., Dewar, M. J. S.: J. Am. Chem. Soc. 100, 6267 (1978)
For instance, see Jug, K., Nanda, D. N.: Theoret. Chim. Acta (Berl.) 57, 107 (1980); Theoret. Chim. Acta (Berl.) 57, 131 (1980)
Riveros, J. M., Wilson, E. B.: J. Chem. Phys. 46, 4605 (1967)
Shimanouchi, T., Matuura, H., Ogawa, Y., Harada, I.: J. Phys. Chem. Ref. Data 9, 1149 (1980).
Kirkwood, J. G.: J. Chem. Phys. 2, 351 (1934)
Janz, G. J., Tomkins, R. P. T.: Nonaqueous Electrolytes Handbook, Vol. I., Academic Press 1972
Nace, H. R.: J. Am. Chem. Soc. 81, 5428 (1959)
They are available upon request to the authors
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ishida, K., Mayama, S. Intrinsic reaction coordinate for the gas-phase pyrolysis of ethyl formate. Theoret. Chim. Acta 62, 245–255 (1983). https://doi.org/10.1007/BF00548838
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00548838