Abstract
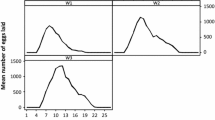
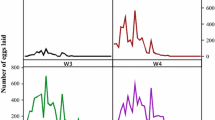
The development and infection dynamics of Babesia ovis in the hemolymph, ovaries, and eggs of Rhipicephalus bursa are described quantitatively, based mainly on examination of Giemsastained smears. After alimentary infection of female ticks, their hemolymph became infected 5 days after repletion (p.repl.). The prevalence and mean intensity of infection increased during the course of infection studied, up to 17 days p.repl. After vertical infection of female ticks, their hemolymph was infected only during the first 3 days after the onset of infestation (p.infest.) and again after the onset of alimentary infection 5 days p.repl. There was a positive correlation between prevalence and mean intensity of infection in the hemolymph. The prevalence of infection decreased with aging of the unfed adult ticks. After alimentary infection, the ovaries became infected 6 days p.repl., and after vertical infection, 3 days p.infest; they remained infected until the death of the tick. Ticks selected for susceptibility during 18 and 19 vertically infected generations were more susceptible than ticks in their first to third vertically infected generations or alimentarily infected ticks. Eggs deposited on day 1 of oviposition were noninfected after alimentary infection of the female tick. After vertical infection of the tick, even such eggs became infected; the infection, then, was detectable in eggs produced throughout the oviposition period regardless of the infection mode. Intense hemolymph infections induced an increase of egg degeneration and a decrease of total as well as infected egg production. There was a positive correlation between the number of deposited and infected eggs as well as between prevalence and mean intensity of infection in eggs. Alimentary infection contracted from a refractory host was rare (3%). Vertical infection passed to the next generation in 83%–95% of the female ticks. Hemolymph infection of a female tick during the oviposition period indicated alimentary infection. Ovarian infection of a female tick before the onset of alimentary infection or infection of eggs deposited before the onset of alimentary infection indicated vertical infection.
Similar content being viewed by others
References
Abramov IV (1957) Importance of Hyalomma plumbeum Panzer, 1795, as reservoir of Piroplasma caballi, Nuttall and Strickland, 1919. Trudy Vses Inst Eksp Vet 21:241–245 (in Russian)
Abramov IV, Stepanova NJ (1952) Detection of Haemosporidia in eggs of Rhipicephalus bursa. Trudy Vses Inst Eksp Vet 19:56–58 (in Russian)
Brumpt E (1937) Cycle évolutif de Piroplasma canis chez les Ixodinés. C R Soc Biol 124:928–931
Budnik VS (1941) Prolonged maintenance and transovarial transmission of Piroplasma caballi by Dermacentor silvarum. Veterinariya 20 (2):15 (in Russian)
Budnik VS (1960) Efficiency of the microovoscopy method of diagnosing the causative agent of equine piroplasmosis in Dermacentor marginatus. Sb Nauchn Rab Sarat Nauchno-Issled Vet Stn 4:92–97 (in Russian)
Büscher G (1975) Untersuchungen zur Entwicklungsdynamik von Babesia ovis (Piroplasmea) in Ovar and Haemolymphe weiblicher Zecken (Rhipicephalus bursa) (Ixodoidea). Veterinary Medicine Dissertation, Veterinary School Hannover
Büscher G (1988) The infection of various tick species with Babesia bigemina, its transmission and identification. Parasitol Res 74:324–330
Callow LL (1965) Babesia bigemina in ticks grown on nonbovine hosts and its transmission to these hosts. Parasitology 55:375–381
Conover WJ (1980) Practical nonparametric statistics, 2nd edn. John Wiley & Sons, New York
Donnelly J, Peirce MA (1975) Experiments on the transmission of Babesia divergens to cattle by the tick Ixodes ricinus. Int J Parasitol 5:363–367
D'yakonov LP (1970) Development of Babesia ovis (Babes, 1892) in ticks and in vertebrate hosts. Trudy Vses Inst Eksp Vet 38:27–35 (in Russian)
Enigk K (1944) Weitere Untersuchungen zur übertrÄgerfrage der Pferdepiroplasmose. Arch Wiss Prakt Tierheilkd 79:58–80
Friedhoff KT (1969) Lichtmikroskopische Untersuchungen über die Entwicklung von Babesia ovis (Piroplasmidea) in Rhipicephalus bursa (Ixodoidea): I. Die Entwicklung in weiblichen Zecken nach der Repletion. Z Parasitenkd 32:191–219
Friedhoff KT (1970) Studies on the life cycle of Babesia ovis. J Parasitol 56 (4) II 1:109–110
Friedhoff KT, Scholtyseck E (1968) Feinstrukturen von Babesia ovis (Piroplasmidea) in Rhipicephalus bursa (Ixodoidea): Transformation sphÄroider Formen zu Vermiculaformen. Z Parasitenkd 30:347–359
Gasanov AA (1963) The infection of Rhipicephalus bursa (Can. et Fanz., 1877) with Babesia ovis in the Nakhichevan Region of the USSR. Izv Akad Nauk Az SSR Ser Biol Med Nauk 4:117–123 (in Russian)
Gray JS, Potgieter FT (1981) The retention of Babesia bigemina infection by Boophilus decoloratus exposed to imidocarb dipropionate during engorgement. Onderstepoort J Vet Res 48:225–227
Holbrook AA, Anthony DW, Johnson AJ (1968) Observations on the development of Babesia caballi (Nuttall) in the tropical horse tick Dermacentor nitens Neumann. J Protozool 15:391–396
KÄhler J (1981) Untersuchungen zur ImmunogenitÄt der Entwicklungsstadien von Babesia ovis aus dem Ovar von Rhipicephalus bursa. Veterinary Medicine Dissertation, Veterinary School Hannover
Kartashev MV (1957) Studies on the infection rate of Dermacentor with Piroplasma caballi under different agricultural conditions in the central region of the USSR. Trudy Vses Inst Eksp Vet 27:210–220 (in Russian)
Kartashev MV (1971) Developmental forms of Babesiella ovis in Rhipicephalus bursa eggs. Mater 1 Sezda Vses Obshch Protozool (Baku Oct. 1971), pp 216–217 (in Russian)
Li PN (1956a) A simplified method for the diagnosis of Babesia ovis in the tick Rhipicephalus bursa. Veterinariya 33(5): 70–71 (in Russian)
Li PN (1956b) Material for studying the life cycle of Babesiella ovis in the vector ticks, Rhipicephalus bursa. Trudy 2 Nauchn Konf Parazit USSR Kiev, pp 76–78 (in Russian)
Li PN (1958) The interrelationship between the feeding of infected Rhipicephalus bursa ticks and the development of Babesiella ovis in them. Trudy Ukr Nauchno-Issled Inst Eksp Vet 24:271–282 (in Russian)
Li PN (1963) Spontaneous infections of Babesiella ovis in Rhipicephalus bursa in the Crimea. Sb Nauchn Rab Sarat Nauchno-Issled Vet Stn 6:163–168 (in Russian)
MacLeod J (1961) Arthropod transmission of microorganisms. Nature 191:885–888
Mahoney DF, Mirre GB (1977) The selection of larvae of Boophilus microplus infected with Babesia bovis (syn B. argentina) Res Vet Sci 23:126–127
Mahoney DF, Mirre GB (1979) A note on the transmission of Babesia bovis (syn. B. argentina) by the one-host tick, Boophilus microplus. Res Vet Sci 26:253–254
Marchenko GF (1947) On some conditions of preservation and transmission of Piroplasma caballi by Dermacentor silvarum. Sb Nauchn Tr Stavropol Inst Mikrobiol Epidem 1:67–73 (in Russian)
Margolis L, Esch GW, Holmes IC, Kuris AM, Schad GA (1982) The use of ecological terms in parasitology (Report of an ad hoc committee of the American Society of Parasitologists). J Parasitol 68:131–133
Markov AA, Abramov IV (1970) Results of a 20 years' observation of repeated cycles of Babesia ovis in 44 generations of Rhipicephalus bursa. Trudy Vses Inst Eksp Vet 38:5–14 (in Russian)
Martin HM, Barnett SF, Vidler BO (1964) Cyclic development and longevity of Theileria parva in the tick Rhipicephalus appendiculatus. Exp Parasitol 15:527–555
Muangyai M (1974) Quantitative Untersuchungen zur transovarialen Infektion von Boophilus microplus (Ixodoidea) mit Babesia bigemina (Piroplasmea). Veterinary Medicine Dissertation, Veterinary School Hannover
Nikol'sky SN, Prokhorova PK (1972) Ixodid ticks-reservoir of babesiids (Preliminary note). Veterinariya 49 (4):56–58 (in Russian)
Nikol'sky SN, Prokhorova PK, Baturina FM (1975) The infection of animals by for them not specific species of babesiids. Dokl Vses Akad Skh Nauk 8:37–38 (in Russian)
Riek RF (1964) The life cycle of Babesia bigemina (Smith and Kilborne, 1893) in the tick vector Boophilus microplus (Canestrini). Aust J Agric Res 15:802–821
Vos AJ de, Stewart NP, Dalgliesh RJ (1984) The effect of chemotherapy on Babesia bigemina in the tick vector Boophilus microplus. Int J Parasitol 14:249–252
Weber G (1972) Glykolmethacrylat-Einbettung und 1–2-Μm-Schnitt-Technik für Zeckengewebe und ganze Zecken. Z Parasitenkd 40:295–306
Yossef LSEA (1980) Untersuchungen über die Möglichkeit einer vertikalen Infektion von Boophilus microplus mit Babesia bigemina (Piroplasmida). Veterinary Medicine Dissertation, Veterinary School Hannover
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Büscher, G., Friedhoff, K.T. & El-Allawy, T.A.A. Quantitative description of the development of Babesia ovis in Rhipicephalus bursa (hemolymph, ovary, eggs). Parasitol Res 74, 331–339 (1988). https://doi.org/10.1007/BF00539454
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00539454