Summary
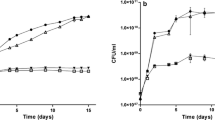
The expression under microaerobic conditions of the Rhizobium meliloti nifA and consequently the nifHDK genes was found to be negatively regulated by ammonia and nitrate. Assimilation of the ammonia to glutamate and glutamine is not required for this regulation to occur. This indicates that ammonia itself, and not a product of its metabolism, may be regulating nif expression. Unlike the situation in Klebsiella pneumoniae, NtrC is apparently not involved in mediating the ammonia effect on nifA expression in R. meliloti. Neither does the fixK gene product, which is known to regulate nifA in R. meliloti, appear to be involved in mediating the ammonia effect. The regulation of nifA by ammonia is shown to be mediated through the FixL protein. A truncated fixJ gene, the product of which has been shown to induce nifA expression irrespective of the oxygen status of the cell, also circumvented the repressive effect of ammonia on nifA expression. This suggests that the ammonia effect is mediated through the FixLJ regulatory cascade. Interstingly no effect of ammonia on fixK expression was observed.
Similar content being viewed by others
References
Batut J, Daveran-Mingot M, David M, Jacobs J, Garnerone AM, Kahn D (1989) fixK a gene homologous with fnr and crp from Escherichia coli regulates nitrogen fixation genes both positively and negatively in Rhizobium meliloti. EMBO J 8:1279–1286
Better M, Ditta G, Helinski DR (1985) Deletion analysis of Rhizobium meliloti symbiotic promoters. EMBO J 4:2419–2424
Birkenhead K, Noonan B, Reville WJ, Boesten B, Manian SS, O'Gara F (1990) Carbon utilization and regulation of nitrogen fixation in Rhizobium meliloti. Mol Plant-Microbe Interact 3:167–173
Boyer HW, Roulland-Dussoix D (1969) A complementation analysis of restriction and modification in Escherichia coli. J Mol Biol 138:179–207
Brewin NJ, Rae AL, Perotto JP, Roberts K, LeGal F, Sindhu SS, Wood EA, Kannenberg EL (1990) Immunological dissection of the plant-microbe interface in pea nodules. In: Gresshoff PM, Roth LE, Stacey G, Newton WE (eds) Nitrogen fixation: Achievements and objectives. Chapman and Hall, New York, London, pp 227–234
de Bruijn FJ, Hilgert U, Stigter S, Schneider M, Meyer H, Klesse U, Pawlowski K (1990) Regulation of nitrogen fixation and assimilation genes in the free-living versus symbiotic state. In Gresshoff PM, Roth LE, Stacey G, Newton WE (eds) Nitrogen fixation: Achievements and objectives. Chapman and Hall, New York, London, pp 33–44
de Bruijn FJ, Rossbach S, Schneider M, Ratet P, Messner S, Szeto WW, Ausubel FM, Schell J (1989) Rhizobium meliloti 1021 has three differentially regulated loci involved in glutamine biosynthesis, none of which is essential for symbiotic nitrogen fixation. J Bacteriol 171:1673–1682
David M, Daveran M-L, Batut J, Dedieu A, Domergue O, Ghai J, Hertig C, Boistard P, Kahn D (1988) Cascade regulation of nif gene expression in Rhizobium meliloti. Cell 54:671–683
De Philip P, Batut J, Boistard P (1990) Rhizobium meliloti FixL is an oxygen sensor and regulates R. meliloti nifA and fixK genes differently in Escherichia coli. J. Bacteriol 172:4255–4262
Ditta G, Virts E, Palomares A, Kim C-H (1987) The nifA gene of Rhizobium meliloti is oxygen regulated. J Bacteriol 169:3217–3223
Dusha I, Bakos A, Kondorosi A, de Bruijn FJ, Schell J (1989) The Rhizobium meliloti early nodulation genes (nodABC) are nitrogen regulated: Isolation of a mutant strain with efficient nodulation capacity on alfalfa in the presence of ammonium. Mol Gen Genet 219:89–96
Figurski DH, Helinski DR (1979) Replication of an origin-containing derivative of plasmid RK2 dependent on a plasmid function provided in trans. Proc Natl Acad Sci USA 76:1648–1652
Gilles-Gonzalez MA, Ditta GS, Helsinski DR (1991) A haemoprotein with kinase activity encoded by the oxygen sensor of Rhizobium meliloti. Nature 350:170–172
Gussin GN, Ronson CW, Ausubel FM (1986) Regulation of nitrogen fixation genes. Annu Rev Genet 20:567–591
Hertig C, Ly RY, Louarn A-M, Garnerone A-M, David M, Batut J, Kahn D, Boistard P (1989) Rhizobium meliloti regulatory gene fixJ activates transcription of R. meliloti nifA and fixK genes in Escherichia coli. J Bacteriol 171:1736–1738
Kahn D, Ditta G (1991) Modular structure of FixJ: homology of the transcriptional activator domain with the −35 binding domain of sigma factors. Mol Microbiol 5:987–997
Kahn ML, Kraus J, Shatters RG (1987) Bacterial catabolism of nitrogen containing compounds in symbiotic nitrogen fixation. In: O'Gara F, Manian SS, Drevon JJ (eds) Physiological limitations and the genetic improvement of symbiotic nitrogen fixation. Kluwer Academic Publishers, Dordrecht, Boston, London, pp 21–27
Kannenberg EL, Brewin NJ (1989) Expression of a cell surface antigen from Rhizobium leguminosarum 3841 is regulated by oxygen and pH. J Bacteriol 171:4543–4548
Kondorosi A, Svab Z, Kiss GB, Dixon RA (1977) Ammonia assimilation and nitrogen fixation in Rhizobium meliloti. Mol Gen Genet 151:221–226
Lewis TA, Gonzalez R, Botsford JL (1990) Rhizobium meliloti glutamate synthase: cloning and initial characterisation of the glt locus. J Bacteriol 172:2413–2420
Manian SS, O'Gara F (1982) Derepression of ribulose bisphosphate carboxcylase activity in Rhizobium meliloti. FEMS Microbiol Lett 14:95–99
Miller JH (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Noonan B, Birkenhead K, Wang Y-P, Boesten B, Dobson A, O'Gara F (1990) Dicarboxylic acid transport and regulation of nitrogen fixation in Rhizobium meliloti. Biochem Soc Trans 18:359–360
O'Gara F, Shanmugam KT (1976) Regulation of nitrogen fixation by Rhizobia: Export of fixed N2 as NH4 +. Biochim Biophys Acta 437:313–321
Osborne MS, Signer ER (1980) Ammonia assimilation in Rhizobium meliloti. J Bacteriol 143:1234–1240
Streeter J (1988) Inhibition of legume nodule formation and N2 fixation by nitrate. CRC Crit Rev Plant Sci 7:1–23
Streeter J (1989) Estimation of ammonium concentration in the cytosol of soybean nodules. Plant Physiol 90:779–782
Szeto WW, Nixon TB, Ronson CW, Ausubel FM (1987) Identification and characterisation of the Rhizobium meliloti ntrC gene: R. meliloti has separate pathways for activation of nitrogen fixation genes in free-living and symbiotic cells. J Bacteriol 169:1423–1432
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Noonan, B., Motherway, M. & O'Gara, F. Ammonia regulation of the Rhizobium meliloti nitrogenase structural and regulatory genes under free-living conditions: Involvement of the fixL gene product?. Molec. Gen. Genet. 234, 423–428 (1992). https://doi.org/10.1007/BF00538701
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00538701