Abstract
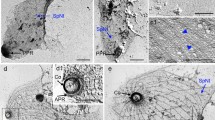
The organization of spines and filaments in whole worms and cytoskeletal fractions of adult Schistosoma mansoni was investigated. The ultrastructure of the spine revealed a closely packed filamentous organization of 3.5- to 5.6-nm elements and electron-lucent areas. Spines were surrounded at the base by electron-dense bodies and membrane invaginations, and covered at the tip by the syncytial surface membrane. Filaments, 7.5–11.1 nm in diameter, were closely associated with the base of the spines, between muscles, near mitochondria or nuclei, and in spaces of the subtegument.
Cytoskeletal fractions prepared by homogenizing adults in Tris-HCl buffer, containing 0.6 M KCl and 1.0% Triton X-100, represented 19%–25% and 32%–38% of wet weight of males and females, respectively. The fractions contained nuclei, spines, 8 to 11 nm filaments, myofibrils, and granules. Vitellaria and egg shells were abundant in fractions from females. Six polypeptides with estimated molecular weights of 130, 96, 84, 78, 74 and 43 kdaltons were identified by sodium dodecyl sulfate-polyacrylamide gel electrophoresis as the major components of the cytoskeleton.
Monoclonal antibody to chicken actin (MAA) was localized predominantly in surface spines and tubercles of adult schistosomes by the indirect immunofluorescence test, while immune serum from infected mice reacted less specifically with the tegument. A 43-kdalton polypeptide with electrophoretic mobility identical to that of vertebrate actin, identified in cytoskeletal and tegumental fractions of adult worms, reacted positively with MAA on immunoblotting. These observations indicate that schistosome actin has electrophoretic and antigenic properties similar to those of vertebrates and that it is detectable in the surface spines of these worms. This study also describes intermediate-sized filaments in an invertebrate and discusses the significance of the spine-filament organization.
Similar content being viewed by others
References
Bennett JL, Seed JL (1977) Characterization and isolation of concanavalin A binding sites from the epidermis of Schistosoma mansoni. J Parasitol 63:250–258
Bogitsh BJ (1982) Schistosoma mansoni: cytochemistry and morphology of the gastrodermal Golgi apparatus. Exp Parsitol 53:57–67
Brown SW, Levinson W, Spudich JA (1976) Cytoskeletal elements of chick embryo fibroblasts revealed by detergent extraction. J Supramol Struct 5:119–130
Cohen C, Reinhardt B, Castellani L, Norton P, Stirewalt M (1982) Schistosome surface spines are “crystals” of actin. J Cell Biol 95:987–988
David S, Smith MA, Clegg JA (1980) Surface proteins of Schistosoma mansoni and their expression during morphogenesis. Eur J Immunol 10:573–575
Davis AH, Blanton R, Klich P (1985) Stage and sex specific differences in actin gene expression in Schistosoma mansoni. Mol Biochem Parasitol 17:289–298
Hockley DJ (1973) Ultrastructure of the tegument of Schistosoma. In: Dawes B (ed) Advances in Parasitology Vol 11, Academic Press New York, p 233
Kusel JR (1972) Protein composition and protein synthesis in the surface membranes of Schistosoma mansoni. Parasitology 65:55–69
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 277:680–685
Lazarides E (1980) Intermediate filaments as mechanical integrators of cellular space. Nature 283:249–256
Lawrence JD (1973) The ingestion of red blood cells by Schistosoma mansoni. J Parasitol 59:60–63
Morris GP (1971) The fine structure of the tegument and associated structures of the cercaria of Schistosoma mansoni. Z Parasitenkd 36:15–31
Norden AP, Aronstein WS, Strand M (1982) Schistosoma mansoni: identification, characterization and purification of the spine glycoprotein by monoclonal antibody. Exp Parasitol 54:432–442
Oaks JA, Cain GD, Mower DA, Raj RK (1981) Disruption and removal of the tegument from Schistosoma mansoni with Triton X-100. J Parasitol 67:761–775
Oaks JA, Cain GD, Mower DA, Raj RK (1983) Comparison of calcium, freeze-thaw and triton X-100 tegumental disruption/recovery techniques applied to Schistosoma mansoni. J Parasitol 69:519–533
Osborn M, Weber K (1977) The detergent-resistant cytoskeleton of tissue cultured cells includes the nucleus and the microfilament bundles. Exp Cell Res 106:339–349
Osborn M, Weber K (1982) Intermediate filaments: cell-type-specific markers in differentiation and pathology. Cell 31:303–306
Pearson AGM, Fincham AG, Waters H, Bundy DAP (1985) Differences in composition between Fasciola hepatica spines and cestode hooks. Comp Biochem Physiol 81B:373–376
Samuelson JC, Caulfield JP, David JR (1982) Schistosomula of Schistosoma mansoni clear concanavalin A from their surface by sloughing. J Cell Biol 94:355–362
Simpson AJG, Schryer MD, Cesari IM, Evans WH, Smithers SR (1981) Isolation and partial characterization of the tegumental outer membrane of adult Schistosoma mansoni. Parasitology 83:163–177
Smith SR, Reynolds ES, von Lichtenberg F (1969) The integument of Schistosoma mansoni. Am J Trop Med Hyg 18:28–49
Studier FS (1973) Analysis of bacteriophage T7 early RNAs and proteins on slab gels. J Mol Biol 79:237–248
Taylor DW, Wells PZ (1984) Isolation and antigen analysis of surface tegument membranes from schistosomula of Schistosoma mansoni. Parasitology 89:495–510
Wilson RA, Barnes PE (1979) Synthesis of macromolecules by the epithelial surfaces of Schistosoma mansoni: an autoradiographic study. Parasitology 78:295–310
Wray W, Boulikas T, Wray VP, Hancock R (1981) Silver staining of proteins in polyacrylamide gels. Anal Biochem 118:197–203
Wu Y, Parker LM, Binder NE, Beckett MA, Sinard JH, Griffiths CT, Rheinwald JG (1982) The mesothelial keratins: new family of cytoskeletal proteins identified in cultured mesothelial cells and non-keratinizing epithelia. Cell 31:693–703
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Abbas, M.K., Cain, G.D. Actin and intermediate-sized filaments of the spines and cytoskeleton of Schistosoma mansoni . Parasitol Res 73, 66–74 (1987). https://doi.org/10.1007/BF00536338
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00536338