Abstract
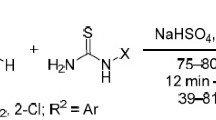
The action of potassium salts of cyanoamino-symm-triazines on esters of chloroacetic and bromomalonic acids gave carbonylalkylamino-symm-triazines. The solvolysis of these esters proceeds with heterocyclilzation, leading to the formation of hydantoin derivatives.
Similar content being viewed by others
References
V. V. Dovlatyan, É. N. Ambartsumyan, and L. A. Khachatryan, Arm. Khirn. Zh., 33, 311 (1980).
V. V. Dovlatyan, L. A. Khachatryan, and É. N. Ambartsumyan, Arm. Khim. Zh., 35, 684 (1982).
V. V. Dovlatyan, L. A. Khachatryan, and É. N. Ambartsumyan, Arm. Khim. Zh., 35, 799 (1982).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 8, pp. 1114–1116, August, 1993.
Rights and permissions
About this article
Cite this article
Dovlatyan, V.V., Ambartsumyan, É.N., Gyul'budagyan, L.L. et al. Synthesis of N-cyano-N-alkoxycarbonylalkylamino-symm-triazines and their solvolysis. Chem Heterocycl Compd 29, 949–951 (1993). https://doi.org/10.1007/BF00534276
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00534276