Abstract
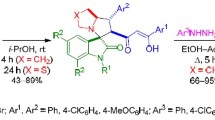
Reaction of N-(1-pyrrolidinyl)-, N-(1-piperidinyl)-, N-(4-morpholinyl)-, and N-(1-hexahydroazepinyl)-thioureas with propiolic acid gives hetero-N-spiro-4′-(2′-amino-Δ2-1′,3′,4′-thiadizolin-4′-io-5′-acetates), which rearrange in acetic anhydride to 2-(N-heteryl)imino-2,3-dihydro-4H-1,3-thiazin-4-ones.
Similar content being viewed by others
References
V. Ya. Kauss, A. F. Mishnev, and I. Ya. Kalvin'sh, Khim. Geterotsikl. Soedin., No. 12, 1674 (1992).
K. A. Jensen, U. Anthoni, B. Kagi, Ch. Larsen, and C. Pedersen, Acta Chem. Scand., 22, No. 1, 1 (1968).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 2, pp. 263–265, February, 1993.
Rights and permissions
About this article
Cite this article
Kauss, V.Y., Kalvin'sh, I.Y. Spirocyclic betaines or 2-amino-Δ2-1,3,4-thiadiazoline-5-acetic acid and their rearrangement in acetic anhydride. Chem Heterocycl Compd 29, 231–233 (1993). https://doi.org/10.1007/BF00531673
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00531673