Abstract
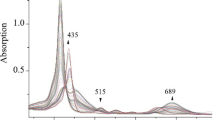
The mass spectral behavior of functionally substituted meso(p-adamantylphenyl)triphenylporphyrins and their p-methoxy-substituted analogs was studied. Breakdown of the M+ ions of these compounds proceeds chiefly-with a loss of the functional substituent in the adamantane ring with the formation of ions, which make it possible to determine the nature of this substituent.
Similar content being viewed by others
References
I. M. Karnaukh, A. S. Moskovkin, and G. V. Ponomarev, Khim. Geterotsikl. Soed., No. 11, 1478 (1993).
G. V. Ponomarev, L. N. Lavrova, A. M. Shul'ga, A. S. Moskovkin, and I. M. Karnaukh, Khim. Geterotsikl. Soed., No. 10, 1428 (1991).
G. V. Ponomarev, V. P. Suboch, and A. N. Lyashko, Khim. Geterotsikl. Soed., No. 6, 773 (1978).
A. D. Adler, J. H. Green, and M. Mautner, Org. Mass Spectrom., 3, 955 (1970).
M. Meot-Ner, A. D. Adler, and J. H. Green, Org. Mass Spectrom., 9, 72 (1974).
P. A. Sharbatyan, P. B. Terent'ev, V. B. Kovalev, and É. A. Shokova, Zh. Org. Khim., 16, 308 (1980).
K. M. Smith (ed.), Porphyrins and Metalloporphyrins, Elsevier Scien. Publ. Comp., Amsterdam (1975), p. 381.
N. S. Vul'fson, V. G. Zaikin, and A. I. Mikaya, Mass Spectrometry of Organic Compounds [in Russian], Khimiya, Moscow (1986), p. 184.
Author information
Authors and Affiliations
Additional information
For Communication 28, see [1].
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 12, pp. 1642–1644, December, 1993.
Rights and permissions
About this article
Cite this article
Karnaukh, I.M., Moskovskin, A.S. & Ponomarev, G.V. Porphyrins. 29. Mass spectra of functionally substituted tetraarylporphyrin derivatives. Chem Heterocycl Compd 29, 1418–1420 (1993). https://doi.org/10.1007/BF00531403
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00531403