Abstract
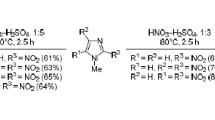
The reactions of 4,5-dinitroimidazole and 5(4)-nitroimidazole-4(5)-sulfonic acid with nucleophilic agents were studied. Mercapto-, alkoxy-, and aminonitroimidazoles were synthesized. In the reaction of dinitroimidazole with sodium alkoxides 5(4)-nitroimidazole was obtained in addition to alkoxynitroimidazoles. It is shown that in the formation of salts of the starting imidazoles with bases nucleophilic-substitution reactions take place only with “soft” reagents.
Similar content being viewed by others
Literature cited
P. M. Kochergin, Khim. Geterotsikl. Soedin., No. 5, 749 (1966).
Y. E. Balaban, J. Chem. Soc., 268 (1930).
R. G. Fargher and F. L. Ryman, J. Chem. Soc., 115, 217 (1919).
V. S. Mokrushin, I. S. Selezneva, T. A. Pospelova, V. K. Usova, S. A. Malinskaya, G. M. Anoshina, T. É. Zubova, and Z. V. Pushkareva, Khim.-farm. Zh., No. 3, 303 (1982).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheakikh Soedinenii, No. 6, pp. 808–810, June, 1983.
Rights and permissions
About this article
Cite this article
Mokrushin, V.S., Belyaev, N.A., Kolobov, M.Y. et al. Reactions of 4,5-dinitroimidazole and 4(5)-nitroimidazole-5(4)-sulfonic acid with nucleophiles. Chem Heterocycl Compd 19, 650–652 (1983). https://doi.org/10.1007/BF00523079
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00523079