Abstract
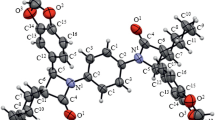
2,2′-Dipropionylazoxybenzenes undergo rearrangement to the corresponding 3-[1-alkoxy-1-(2-propionylarylamino)ethyl]benzo[c]isoxazoles in the case of base catalysis; this transformation is realized only in the case of ortho,ortho' orientation of the propionyl groups in the substrate molecule and with the participation of the solvent as a reagent. A mechanism for the rearrangement that assumed the participation of one of the propionyl groups in intramolecular reduction of the azoxy group to an azo group is proposed.
Similar content being viewed by others
Literature cited
S. S. Mochalov, A. N. Fedotov, and Yu. S. Shabarov, Zh. Org. Khim., 16, 462 (1980).
S. S. Mochalov, A. N. Fedotov, and Yu. S. Shabarov, Zh. Org. Khim., 15, 947 (1979).
S. S. Mochalov, A. N. Fedotov, E. A. Kupriyanova, and Yu. S. Shabarov, Khim. Geterotsikl. Soedin., No. 5, 688 (1983).
R. Y. Ning, J. F. Blount, W. Y. Chen, and P. M. Madan, J. Org. Chem., 40, 2201 (1973).
V. Nelson, A. Serianz, and P. Kovacic, J. Org. Chem., 41, 1751 (1976).
B. T. Newbold, J. Org. Chem., 27, 3919 (1962).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 6, pp. 743–748, June, 1983.
Rights and permissions
About this article
Cite this article
Mochalov, S.S., Fedotov, A.N. & Shabarov, Y.S. Rearrangement of 2,2′-dipropionylazoxybenzenes to 3-[1-alkoxy-1-(2-propionylarylamino)ethyl]benzo[c]isoxazoles. Chem Heterocycl Compd 19, 593–598 (1983). https://doi.org/10.1007/BF00523066
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00523066