Abstract
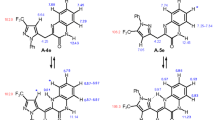
The chlorination of 4-phenyl-2,3-dihydro-1H-1,5-benzodiazepin-2-one with N-chlorosuccinimide takes place at the methylene group to give mono and diehloro derivatives. In the reaction of the diazepinone with sulfuryl chloride chlorine is incorporated in the 1 or 3 position or in both the 1 and 3 positions, as well as in the para position of the phenyl substituent; in the presence of anhydrous aluminum chloride substitution takes place in the methylene group of the heteroring and in the 8 position of the annelated benzene ring.
Similar content being viewed by others
Literature cited
A. V. Bogatskii, R. Yu. Ivanova, and S. A. Andronati, Ref. Zh. Khim., 17, 0129 (1980).
R. Yu. Ivanova, S. A. Andronati, Z. I. Zhilina, and T. A. Voronina, News in the Chemistry of Nitrogen-Containing Heterocycles [in Russian], Vol. 1, Riga (1979), p. 170.
S. Fielding and I. Hoffmann, Br. J. Chem. Pharmacol., No. 7 (Suppl. 1), 7–15 (1979); Chem. Abstr., 91, 4 9408 (1979).
Z. F. Solomko and A. N. Kost, Khim. Geterotsikl. Soedin., No. 11, 1443 (1975).
A. N. Kost, P. A. Sharbatyan, P. B. Terent'ev, Z. F. Solomko, and V. S. Tkachenko, Zh. Org. Khim., 8, 2113 (1972).
T. S. Chmilenko and Z. F. Solomko, Khim. Geterotsikl. Soedin., No. 6, 834 (1977).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 3, pp. 405–409, March, 1982.
Rights and permissions
About this article
Cite this article
Solomko, Z.F., Sheremet, V.I., Khmel', M.P. et al. Chloro derivatives of 4-phenyl-2,3-dihydro-1H-1,5-benzodiazepin-2-one. Chem Heterocycl Compd 18, 307–310 (1982). https://doi.org/10.1007/BF00522136
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00522136