Abstract
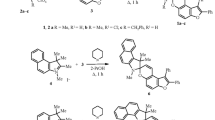
New spirans of the 5,5-dimethyl-3-phenyl-2-oxazolidone series that display photo-chromic properties in alcohol solutions at -80 °C were synthesized. The photoinduced forms are characterized by the presence of two long-wave absorption bands at 350–420 nm and 500–650 nm. The 1H and 13C NMR spectra were studied. Anisochronicity of the diastereotopic methyl groups of the oxazolidone ring shows up only in the 13C NMR spectra.
Similar content being viewed by others
Literature cited
B. Ya. Simkin, S. P. Makarov, N. G. Furmanova, K. Sh. Karaev, and V. I. Minkin, Khim. Geterotsikl. Soedin., No. 6, 747 (1984).
B. Ya. Simkin, V. I. Minkin, and L. E. Nivorozhkin, Khim. Geterotsikl. Soedin., No. 9, 1180 (1978).
J. Hlubeck, R. Rithie, and W. C. Taylor, Aust. J. Chem., 29, 2347 (1971).
A. M. Samat, R. J. Guglielmetti, and G. Martin, J. Org. Magn. Reson., 8, 62 (1976).
G. C. Levy and G. L. Nelson, Carbon-13 Nuclear Magnetic Resonance for Organic Chemists, New York (1972).
D. Cameron Margaret, US Patent No. 2844590; Ref. Zh. Khim., No. 22, 89742 (1960).
N. Donaldson, The Chemistry and Technology of Compounds of the Naphthalene Series [Russian translation], Goskhimizdat, Moscow (1963), p. 317.
G. F. Hennion and A. P. Boiselle, J. Org. Chem., 26, 725 (1961).
Author information
Authors and Affiliations
Additional information
See [1] for communication 15.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 8, pp. 1031–1035, August, 1985.
Rights and permissions
About this article
Cite this article
Luk'yanov, B.S., Nivorozhkin, L.E., Chernysh, Y.E. et al. Photo- and thermochromic spirans. 16. 2-Oxo-3-phenyl-5,5-dimethylspiro(1,3-oxazolidine-4,2′-[2h]chromenes). Chem Heterocycl Compd 21, 857–861 (1985). https://doi.org/10.1007/BF00519808
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00519808