Summary
-
1.
Capsaicin (CAP) has been shown to exert a selective neurotoxic effect on peptidergic primary afferent neurons. The effect of CAP on the isolated guinea-pig left auricle and papillary muscle preparations and on the isolated rat uterus was used to elucidate its mode and site of action with regard to cellular Ca2+ utilization.
-
2.
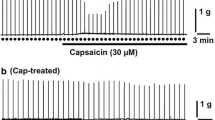
In the electrically driven left auricle CAP first increased and then decreased the size of the contractions while in the electrically driven papillary muscle CAP caused only a decrease in the contractions. Electrophysiological measurements showed that the initial increase in contraction size coincided with a decrease in the upstroke velocity of the action potential. This membrane-stabilizing effect of CAP seemed also responsible for the decrease in contractile activity.
-
3.
The positive inotropic effect of CAP on the left auricle was concentration-dependent (0.03–6.5 μM). The positive inotropic effect of 0.33 μM CAP was reproducible at intervals of 15 min, whereas tachyphylaxis developed at shorter intervals or higher concentrations of CAP. The percent increase in the size of contractions by 0.33 μM CAP was smaller when [Ca2+]e was double but larger when 2.2 μM verapamil or 0.1 mM La3+ was present. The increase in contractions by 0.4–400 μM isoproterenol was greatly reduced by 0.33 μM CAP in a noncompetitive manner. The positive inotropic effect of 2.9 μM glucagon was also inhibited by 0.33 μM CAP.
-
4.
In the isolated anoestrous rat uterus 0.03–3.3 μM CAP caused a transient inhibition of the spontaneous contractions similarly to the effect of 2.2 μM verapamil.
-
5.
It is concluded that the positive inotropic effect of CAP in the left auricle may be due to an increase in [Ca2+]i which does not result from a transmembraneous influx of Ca2+ but probably from a release of Ca2+ from stores inaccessible to either verapamil and La3+. The negative inotropic effect of CAP and the inhibition of the positive inotropic effects of isoproterenol and glucagon may be explained by an unspecific membrane-stabilizing effect of CAP. A membrane-stabilizing effect may also be responsible for the CAP-induced inhibition of the spontaneous contractions of the rat uterus.
Similar content being viewed by others
References
Bailey LE, Fawzi AB (1982) The role of superficially bound calcium in the regulation of myocardial contractility and the positive inotropic response to ouabain. Can J Physiol Pharmacol 60: 568–575
Durrett LR, Adams HR (1980) A comparison of the influence of La3+, D600 and gentamycin on frequency-force relationships in isolated myocardium. Eur J Pharmacol 66: 315–3225
Fabiato A, Fabiato F (1979) Calcium and cardiac excitation-contraction coupling. Ann Rev Physiol 41: 473–484
Fukuda N, Fujiwara M (1969) Effect of capsaicin on the guinea-pig isolated atrium. J Pharm Pharmacol 21: 622–624
Gamse R, Molnar A, Lembeck F (1979) Substance P release from spinal cord slices by capsaicin. Life Sci 25: 629–636
Honerjäger P, Loibl E, Ulm K (1983) Does sodium channel blockade explain the negative inotropic effect of class 1 antiarrhythmic drugs? Naunyn-Schmiedeberg's Arch Pharmacol 322 [Suppl]: R30
Jancsó G, Kiraly E, Karcsu S, Szebeni A, Toth L, Bacsy E, Joo F, Parducz A (1984) Neurotoxin induced nerve cell degeneration: possible involvement of calcium. Brain Res (in press)
Lembeck F, Gamse R (1982) Substance P in peripheral sensory processes. In: Ciba Foundation symposium 91, Substance P in the nervous system. Pitman, London, pp 35–54
Lundberg JM, Hua X, Fredholm BB (1984) Capsaicin-induced stimulation of the guinea-pig atrium. Involvement of a novel transmitter or a dirrect action on myocytes? Naunyn-Schmiedeberg's Arch Pharmacol 325: 176–182
Miller MS, Buck SS, Sipes IG, Yamamura HI, Burks TF (1982) Regulation of substance P by nerve growth factor: disruption by capsaicin. Brain Res 250: 193–196
Molnar J, György L, Unyi G, Kenyeres J (1969) Effect of capsaicin on the isolated ileum and auricle of the guinea pig. Acta Physiol Acad Sci Hung 35: 369–374
Nagy JI (1982) Capsaicin: a chemical probe for sensory neuron mechanisms. In: Iversen LL, Iversen SD, Snyder SH (eds) Handbook of psychophyarmacology, vol 15. Plenum, New York, pp 185–235
Nawrath H (1981) Action potential, membrane currents and force of contraction in mammalian heart muscle fibers treated with quinidine. J Pharmacol Exp Ther 216: 176–182
Nayler WG (1975) Some factors which influence the amount of calcium stored at the superficially located sites in cardiac muscle cells. In: Fleckenstein A, Dhalla NS (eds) Recent advances in studies on cardiac structure and metabolism, vol 5. University Park Press, Baltimore, pp 73–79
Ravens U (1975) The effects of lanthanum on electrical and mechanical events in mammalian cardiac muscle. Naunyn-Schmiedeberg's Arch Pharmacol 288: 133–146
Saria A, Skofitsch G, Lembeck F (1982) Distribution of capsaicin in rat tissues after systemic administration. J Pharm Pharmacol 34: 273–275
Toda N, Usui H, Nishino N, Fujiwara M (1972) Cardiovascular effects of capsaicin in dogs and rabbits. J Pharmacol Exp Ther 181: 512–521
Tritthart HA (1980) Pharmacology and electrophysiology of calcium ion antagonists. Clin Invest Med 3: 1–7
Williams JT, Zieglgänsberger W (1982) The acute effects of capsaicin on rat primary afferents and spinal neurons. Brain Res 253: 125–131
Windisch H, Tritthart HA (1981) Calcium ion effects on the rising phase of action potentials obtained from guinea-pig papillary muscles at different potassium concentrations. J Mol Cell Cardiol 13: 457–469
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zernig, G., Holzer, P. & Lembeck, F. A study of the mode and site of action of capsaicin in guinea-pig heart and rat uterus. Naunyn-Schmiedeberg's Arch. Pharmacol. 326, 58–63 (1984). https://doi.org/10.1007/BF00518779
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00518779