Abstract
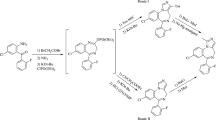
The preparative synthesis of 1H-pyrrolo[2,3-f]isoquinoline is proposed. Electrophilic substitution reactions, viz., the Mannich and Vilsmeier reactions and diazo coupling, were studied for the first time in the pyrroloisoquinoline series.
Similar content being viewed by others
Literature cited
A. P. Gryaznov, R. N. Akhvlediani, T. A. Volodina, A. M. Vasil'ev, T. A. Babushkina, and N. N. Suvorov, Khim. Geterotsikl. Soedin., No. 3, 369 (1977).
T. R. Covindachari and V. Rajappa Sudarsanam, Indian J. Chem., 4, 118 (1966).
T. R. Covindachariand V. Sudarsanam, Indian J. Chem., 5, 16 (1967).
T. R. Covindachari and V. Sudarsanam, Indian J. Chem., 7, 402 (1971).
C. G. Le Fevre and R. J. W. Le Fevre, J. Chem. Soc., No. 10, 1470 (1935).
H. F. Manske and M. Kulka, Can. J. Res., 27B, 161 (1949).
C. Kinast and L.-F. Teitze, Angew. Chem., 88, 261 (1976).
A. A. Polyakova and R. A. Khmel'nitskii, Mass Spectrometry in Organic Chemistry [in Russian], Khimiya, Leningrad (1972), p. 266.
Author information
Authors and Affiliations
Additional information
See [1] for communication 2.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 4, pp. 490–494, April, 1984.
Rights and permissions
About this article
Cite this article
Ponasenkova, T.F., Akhvlediani, R.N., Kurkovskaya, L.N. et al. Pyrroloquinolines and pyrroloisoquinolines. 3. Synthesis of 1H-pyrrolo[2,3-f]isoquinoline and some of its derivatives. Chem Heterocycl Compd 20, 394–398 (1984). https://doi.org/10.1007/BF00513853
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00513853