Abstract
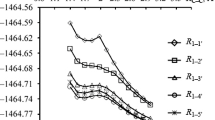
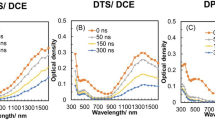
Dimers of 4(5)-phenyl-5(4)-β-naphthyl-2-phenyl-substituted imidazolyl radicals were synthesized in order to study the effect of a β-naphthyl group on the stability of triaryl-imidazolyl radicals, and the rate constants and energies of activation for dissociation of the dimers were determined. The presence of a β-naphthyl group in the bistriarylimid-azolyl molecule facilitates its dissociation into radicals as compared with bistriphenyl-imidazolyl.
Similar content being viewed by others
Literature cited
B. S. Tanaseichuk, A. A. Bardina, and V. A. Maksakov, Zh. Organ. Khim., 7, 1508 (1971).
A. A. Bardina, B. S. Tanaseichuk, and A. A. Khomenko, Zh. Organ. Khim., 7, 1267 (1971).
T. S. Tanaseichuk, K. V. Stanovkina, A. N. Sunin, and L. G. Rezepova, Zh. Organ. Khim., 5, 2054 (1969).
B. S. Tanaseichuk and L. G. Rezepova, Zh. Organ. Khim., 6, 1065 (1970).
D. M. White and J. Sonnenberg, J. Am. Chem. Soc., 88, 3825 (1966).
S. Goldschmidt and K. Renn, Ber., 55, 628 (1922).
Author information
Authors and Affiliations
Additional information
See [1] for communication XIII.
Translated from Khimiya Geterotsiklicheskikh Soedinnii, No. 9, pp. 1255–1257, September, 1971.
Rights and permissions
About this article
Cite this article
Tanaseichuk, B.S., Bardina, A.A. & Khomenko, A.A. Investigation of nitrogen-containing heterocyclic free radicals. Chem Heterocycl Compd 7, 1180–1182 (1971). https://doi.org/10.1007/BF00510032
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00510032