Abstract
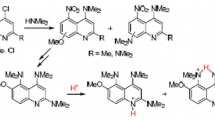
3,3-Dimethyl-2-oxa-1,2,3,4-tetrahydroacridine and its N-oxide and 3,3,6,6-tetramethyl-2,7-dioxa-sym-octahydroacridine N-oxide were synthesized. On heating with acetic anhydride, the N-oxides form, respectively, acetates of 3,3-dimethyl-2-oxa-1,2,3,4-tetrahydro-4-acridinol and 3,3,6,6-tetramethyl-2,7-dioxa-sym-octahydro-4-acridinol. Hydrolysis of the acetates gives the alcohols themselves. Oxidation of 3,3,6,6-tetramethyl-2,7-dioxa-sym-octahydro-4-acridinol acetate gives the corresponding N-oxide. Oxidation of 3,3,6,6-tetramethyl-2,7-dioxa-sym-octahydro-4-acridinol with manganese dioxide gives 3,3,6,6-tetramethyl-2,7-dioxa-sym-octahydro-4-acridinone.
Similar content being viewed by others
Literature cited
G. A. Klimov and M. N. Tilichenko, Zh. Organ. Khim., 1, 977 (1965).
G. A. Klimov and M. N. Tilichenko, Khim. Geterotsikl. Soedin., 306 (1967).
G. A. Klimov, M. N. Tilichenko, and E. S. Karaulov, Khim. Geterotsikl. Soedin., 297 (1969).
S. N. Golikov and V. I. Rozengart, Cholinesterases and Anticholinesterase Substances [in Russian], Meditsina (1964), p. 133.
E. Bamberger and E. Demuth, Ber., 34, 1330 (1901).
A. N. Elizarova and I. N. Nazarov, Izv. Akad. Nauk SSSR, Otd. Khim. Nauk, 223 (1940).
M. N. Tilichenko, Izv. Vuzov, Ser. Khim., 4, 96 (1961).
Yu. V. Karyakin and I. I. Angelov, Pure Chemical Reagents [in Russian], Moscow (1955), p. 333.
V. A. Stonik, V. I. Vysotskii, and M. N. Tilichenko, Khim. Geterotsikl. Soedin., 1542 (1970).
Author information
Authors and Affiliations
Additional information
See [9] for communication VII.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 9, pp. 1218–1221, September, 1971.
Rights and permissions
About this article
Cite this article
Klimov, G.A., Tilichenko, M.N. & Timofeeva, L.A. Hydroacridines and related compounds. Chem Heterocycl Compd 7, 1145–1148 (1971). https://doi.org/10.1007/BF00510024
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00510024