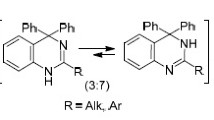
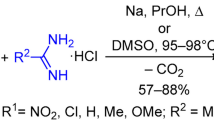
Abstract
Monosubstituted benzamidines undergo cyclization with quinoxalinum salts to give 1,2-disubstituted 3a,4,9,9a-tetrahydroimidazo[4,5-b]quinoxalines. The participation in similar cyclizations of N,N′-disubstituted amidines with a CH-active group in the a position leads to 3a,4,9,9a-tetrahydropyrrolo[2,3-b]quinoxalines.
Similar content being viewed by others
Literature cited
V. N. Charushin, V. G. Baklykov, O. N. Chupakhin, and L. M. Naumova, Khim. Geterotsikl. Soedin., No. 9, 1284 (1984).
O. N. Chupakhin, V. N. Charushin, L. M. Naumova, A. I. Rezvukhin, and N. A. Klyuev, Khim. Geterotsikl. Soedin., No. 11, 1549 (1981).
S. Patai (editor), The Chemistry of Functional Groups. The Chemistry of Amidines and Imidates, Wiley, London (1975).
V. N. Charushin, V. G. Baklykov, O. N. Chupakhin, N. N. Vereshchagina, L. M. Naumova, and N. N. Sorokin, Khim. Geterotsikl. Soedin., No. 12, 1684 (1983).
V. N. Charushin, V. G. Baklykov, O. N. Chupakhin, G. M. Petrova, and E. O. Sidorov, Khim. Geterotsikl. Soedin., No. 5, 680 (1984).
V. G. Baklykov, V. N. Charushin, O. N. Chupakhin, and V. N. Drozd, Khim. Geterotsikl. Soedin., No. 5, 686 (1984).
P. Oxley, H. W. Partrige, and W. Short, J. Chem. Soc., No. 8, 1110 (1947).
Author information
Authors and Affiliations
Additional information
See [1] for Communication 13.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 3, pp. 390–395, March, 1985.
Rights and permissions
About this article
Cite this article
Naumova, L.M., Charushin, V.N., Chupakhin, O.N. et al. Cyclization of N-alkylazinium cations with bifunctional nucleophiles. 14. Reaction of quinoxalinium salts with mono- and N,N′-disubstituted amidines. Chem Heterocycl Compd 21, 326–331 (1985). https://doi.org/10.1007/BF00506676
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00506676