Abstract
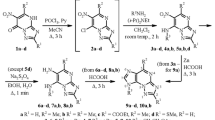
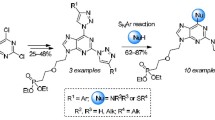
A general method for the synthesis of optically active 9-purinyl-α-amino acids by condensation of 5-amino-4,6-dichloropyrimidine with α,ω-diamino carboxylic acids and subsequent cyclization of the Nω-(5-amino-4-chloro-6-pyrimidinyl)-amino acids with triethyl orthoformate was developed. A number of 6-substituted α-amino-ω-(9-purinyl) carboxylic acids were obtained by nucleophilic substitution of the chlorine atom in the α-amino-ω-(6-chloro-9-purinyl) carboxylic acids.
Similar content being viewed by others
Literature cited
M. Yu. Lidak, Ya. Ya. Shluke, and Yu. P. Shvachkin, Khim. Geterotsikl. Soedin., No. 5, 955 (1968).
A. J. H. Nollet and U. K. Pandit, Tetrahedron, 25, 5983 (1969).
A. J. H. Nollet, C. M. Huting, and U. K. Pandit, Tetrahedron, 25, 5971 (1969).
M. T. Doel, A. S. Jones, and N. Taylor, Tetrahedron Lett., No. 27, 2285 (1969).
J. K. MacLeod, R. E. Summons, C. W. Parker, and D. S. Letham, Chem. Commun., No. 19, 809 (1975).
B. Z. Simkhovich and M. Yu. Lidak, Materials from the 5th Biochemical Conference of the Baltic Republics and Belorussian SSR [in Russian], Tallin (1976), p. 201.
M. Yu. Lidak, Ya. Ya. Shluke, S. E. Poritere, and Yu. P. Shvachkin, Khim. Geterotsikl. Soedin., No. 3, 427 (1971).
Yu. P. Shvachkin, N. A. Voskova, G. A. Korshunova, Yu. A. Semiletova, and N. V. Savelova, Vestn. Mosk. Gos. Univ., Ser. Khim., No. 17, 598 (1976).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 12, pp. 1690–1695, December, 1982.
Rights and permissions
About this article
Cite this article
Poritere, S.E., Shluke, Y.Y., Lidak, M.Y. et al. Synthesis of optically active 9-purinyl-α-amino acids. Chem Heterocycl Compd 18, 1308–1313 (1982). https://doi.org/10.1007/BF00506624
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00506624