Summary
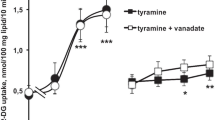
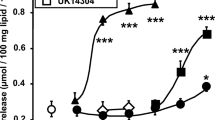
The influence of adenosine on blood flow, lipolysis and adrenergic neurotransmission was studied in canine subcutaneous adipose tissue in situ. Tissue concentrations of endogenous adenosine were elevated in 2 ways: (1) by inhibition of adenosine elimination by i. a. infusion of the uptake blocker dipyridamole (0.5–1.5 μM in arterial plasma) + the deaminase inhibitor erythro-9-(2-hydroxy-3-nonyl)-adenine (EHNA; 3–5μM) or (2) by prolonged nerve stimulation (4 Hz for 30 min). Furthermore, effects of exogenous adenosine and the adenosine antagonist theophylline were studied. Dipyridamole+EHNA increased resting vascular conductance from 0.055 to 0.096 ml×min−1×100 g−1×mm Hg−1, whereas lipolysis induced by sympathetic nerve stimulation (2,4 and 8 Hz during 2 min) was inhibited (by 91, 71 and 34%, respectively). “Therapeutic” concentrations of theophylline (5–15 μg/ml plasma) completely reversed the effects of dipyridamole + EHNA on blood flow and significantly reversed the antilipolytic effect of these drugs. In untreated adipose tissue theophylline reduced resting vascular conductance by 40% and enhanced the lipolytic response to prolonged nerve stimulation but not that to brief nerve stimulation. Transmitter release, evaluated by the outflow of endogenous noradrenaline and/or by the outflow of radioactivity after 3H-noradrenaline labelling, was linearly related to the stimulation frequency (2,4 and 8 Hz). Dipyridamole + EHNA increased plasma noradrenaline by inhibiting uptake or binding of noradrenaline to blood cells. However, noradrenaline release induced by nerve stimulation was not significantly affected by dipyridamole + EHNA or by theophylline. It is concluded that a rise in endogenous adenosine concentration caused by drugs or by prolonged sympathetic nerve stimulation inhibits the lipolytic response to nerve stimulation without any effect on noradrenaline release. The results also show that theophylline in a therapeutic dose range is capable of enhancing lipolysis only if endogenous adenosine concentrations are raised.
Similar content being viewed by others
References
Afonso S (1970) Inhibition of coronary vasodilating action of dipyridamole and adenosine by aminophylline in the dog. Circ Res 26:743–752
Arch JRS, Newsholme EA (1978) The control of the metabolism and the hormonal role of adenosine. In: Aldrich WN, Campbell PW (eds) Essays in biochemistry, vol 14. Academic Press, New York, p 83
Banaschak H, Bluth R (1978) Uptake of noradrenaline by human erythrocytes. Int J Clin Pharmacol 16:336–339
Bretschneider HJ, Frank JA, Bernard U, Kochsiek K, Scheler F (1959) Die Wirkung eines Pyrimidopyrimidinderivates auf die Sauerstoffversorgung des Herzmuskels. Arzneim Forsch 9:49–59
Ebert R, Schwabe U (1973) Studies on the antilipolytic effect of adenosine and related compounds in isolated fat cells. Naunyn-Schmiedeberg's Arch Pharmacol 278:247–259
Fain JN, Malbon CC (1979) Regulation of adenylate cyclase by adenosine. Molec Cell Biochem 25:143–151
Fox IH, Kelley WN (1978) The role of adenosine and 2′ deoxyadenosine in mammalian cells. Ann Rev Biochem 47:655–686
Fredholm BB (1970) Studies on the sympathetic regulation of circulation and metabolism in canine subcutaneous adipose tissue. Acta Physiol Scand Suppl 354:1–47
Fredholm BB (1974) Vascular and metabolic effects of theophylline, dibuturyl cAMP and dibuturyl cGMP in canine subcutaneous adipose tissue in situ. Acta Physiol Scand 90:226–236
Fredholm BB (1976) Release of adenosine-like material from isolated perfused dog adipose tissue following sympathetic nerve stimulation and its inhibition by adrenergic α-receptor blockade. Acta Physiol Scand 96:422–430
Fredholm BB (1978a) Effects of adenosine, adenosine analogues and drugs inhibiting adenosine inactivation on lipolysis in rat fat cells. Acta Physiol Scand 102:191–198
Fredholm BB (1978b) Local regulation of lipolysis in adipose tissue by fatty acids, prostaglandins and adenosine. Medical Biology 56:249–261
Fredholm BB, Rosell S (1970) Fate of 3H-noradrenaline in canine subcutaneous adipose tissue. Acta physiol scand 80:404–411
Fredholm BB, Hedqvist P (1980) Modulation of neurotransmission by purine nucleotides and nucleosides. Biochem Pharmacol 29:1635–1643
Fredholm BB, Sollevi A (1981) The release of adenosine and inosine from canine subcutaneous adipose tissue by nerve stimulation and noradrenaline. J Physiol (in press)
Fredholm BB, Sollevi A, Vernet L, Hedqvist P (1980) Inhibition by dipyridamole of stimulated purine release. Naunyn-Schmiedeberg's Arch Pharmacol Suppl. 313:R18
Hedqvist P, Fredholm BB (1976) Effects of adenosine on adrenergic neurotransmission; Prejunctional inhibition and postjunctional enhancement. Naunyn-Schmiedeberg's Arch Pharmacol 293:217–223
Hjemdahl P, Sollevi A (1978) Antilipolytic effects of adenosine in isolated perifused fat cells. Acta physiol Scand 103:270–274
Hjemdahl P, Daleskog M, Kahan T (1979) Determination of plasma catecholamines by high performance liquid chromatography with electrochemical detection: Comparison with a radioenzymatic method. Life Sci 25:131–138
Laurell S, Tibbling G (1966) An enzymatic fluoremetric micro-method for the determination of glycerol. Clin Chem Acta 13:317–322
Ogilvie RI (1978) Clinical pharmacokinetics of theophylline. Clin Pharmacokinetics 3:267–293
Osswald H (1975) Renal effects of adenosine and their inhibition by theophylline in dogs. Naunyn-Schmiedeberg's Arch Pharmacol 288:79–86
Pedersen AK (1979) Specific determination of dipyridamole in serum by high-performance liquid chromatography. J Chromatography 162:98–103
Rosell S (1966) Release of free fatty acids from subcutaneous adipose tissue in dogs following sympathetic nerve stimulation. Acta Physiol Scand 67:343–351
Sattin A, Rall TW (1970) The effect of adenosine and adenine nucleotides on the cyclic adenosine 3′, 5′-phosphate content of guinea pig cerebral cortex slices. Mol Pharmacol 6:13–23
Schaeffer HJ, Schwender CF (1974) Enzyme inhibitors. 26. Bridging hydrophobic and hydrophilic regions on adenosine deaminase with some 9-(2-hydroxy-3-alkyl)adenines. J Med Chem 17:6–8
Schwabe U, Ebert R, Erbler HC (1975) Adenosine release from fat cells. Effect on cyclic AMP levels and hormone actions. Adv. Cyclic Nucleotide Research, vol 5. Raven Press, New York, p 569
Soldin SJ, Hill JG (1977) A rapid micromethod for measuring theophylline in serum by reverse-phase-high performance liquid chromatography. Clin Biochem 10(2):74–77
Sollevi A (1980) The sensitivity of perifused fat cells to the antilipolytic action of adenosine. Acta Physiol Scand 110:211–213
Sollevi A, Fredholm BB (1981a) The antilipolytic effect of endogenous and exogenous adenosine in canine adipose tissue in situ. Acta Physiol Scand (in press)
Sollevi A, Fredholm BB (1981b) Role of adenosine in adipose tissue circulation. Acta Physiol Scand (in press)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sollevi, A., Hjemdahl, P. & Fredholm, B.B. Endogenous adenosine inhibits lipolysis induced by nerve stimulation without inhibiting noradrenaline release in canine subcutaneous adipose tissue in vivo. Naunyn-Schmiedeberg's Arch. Pharmacol. 316, 112–119 (1981). https://doi.org/10.1007/BF00505303
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00505303