Abstract
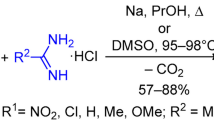
The first step in the acid-catalyzed reaction of 5,6-diamino-1,3-dimethyluracil with carbonyl compounds is the formation of an azomethine at the 5-amino group. Chalcone derivatives undergo a further substitution; the 6-amino group is replaced by a hydroxyl group with subsequent ring closure and the formation of a 2,3-dihydro-1,5-oxazepine ring. Azomethines based on arylidenacetones forms 2,3-dihydropyrimidino[5,6-b]-1,5-diazepine derivatives.
Similar content being viewed by others
Literature cited
V. D. Orlov, I. Z. Papiashvili, and P. A. Grigorov, Khim. Geterosikl. Soedin., No. 5, 671 (1983).
F. Yoneda and M. Higuchi, J. Chem. Soc., Perkin Trans. I, No. 6, 1336 (1977).
S. Wawzonek, J. Org. Chem., 47, 310 (1976).
K. Senga, J. Sato, and S. Nishikaki, Chem. Pharm. Bull., 26, 765 (1978).
G. G. Dang, R. Caugolle, and T. B. Dang, Compt. Rend., 274, 885 (1972).
V. D. Orlov, N. N. Kolos, F. G. Yaremenko, and V. F. Lavrushin, Khim. Geterosikl. Soedin., No. 5, 697 (1980).
V. D. Orlov, S. M. Desenko, and N. N. Kolos, Khim. Geterosikl. Soedin., No. 1, 126 (1984).
J. D. Roberts and C. J. Green, J. Am. Chem. Soc., 68, 214 (1946).
W. Traube and W. Nithack, Ber., 39, 227 (1906).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 2, pp. 241–246, February, 1984.
Rights and permissions
About this article
Cite this article
Orlov, V.D., Papiashvili, I.Z. Reactions of 5,6-diamino-1,3-dimethyluracil with carbonyl compounds. Chem Heterocycl Compd 21, 200–205 (1985). https://doi.org/10.1007/BF00504210
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00504210