Abstract
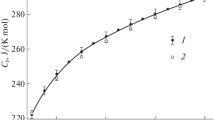
In this paper the authors describe the use of a high-temperature drop calorimeter with autoadiabatic control for the measurment of the enthalpy of α-Al2O3 in the temperature range 550 to 2400 K for both solid and liquid phases. Equations representing the enthalpy of both solid and liquid states are obtained from the data with the use of a computer. In addition to the melting point, T m=2328±7 K, the latent heat of solidification, L=1137.90 J · g−1, has also been determined. The results of the present work are compared with those reported in the literature.
Similar content being viewed by others
References
Y. S. Touloukian, Thermophysical Properties of Matter, Vol. 5 (Plenum, New York, 1970), pp. 24–25.
V. Ya. Chekhovskoi, Eng. Fiz. Zh. 5:62 (1962) (Russian).
V. Ya. Chekhovskoi, Teplofiz. Vysok. Temp. 2:296 (1964) (Russian).
M. Hoch and H. L. Johnston, J. Phys. Chem. 65:1184 (1961).
D. A. Ditmars and T. B. Douglas, J. Res. Natl. Bur. Stand. 75A:401 (1971).
P. H. Zhang, Z. H. Li, R. Z. Chang, and X. N. Zhou, J. Eng. Thermophys. 3:413 (1982) (Chinese).
P. B. Kantor and L. S. Lazareva, Ukr. Fiz. Zh. 7:205 (1963) (Russian).
S. J. Schneider, Pure Appl. Chem. 31:116 (1970).
S. S. Chang, in Proceeding of the Seventh Symposium on Thermophysical Properties, A. Cezairliyan, ed. (Am. Soc. Mech. Eng., New York, 1977), p. 83.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhang, P.H., Chang, R.Z., Wei, Z. et al. The melting point, latent heat of solidification, and enthalpy for both solid and liquid α-Al2O3 in the range 550–2400 K. Int J Thermophys 7, 811–819 (1986). https://doi.org/10.1007/BF00503838
Issue Date:
DOI: https://doi.org/10.1007/BF00503838