Abstract
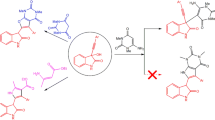
A method for the synthesis of ftorafur metabolites, viz., 3′-hydroxy- and 4′-hydroxyftorafur, from 3-benzoxytetrahydrofuran, which is chlorinated in the presence of UV irradiation, was developed. The resulting mixture of α-chloro ethers is alkylated by 2,4-bis(trimethylsilyl)-5-fluorouracil. The alkylation products are separated by fractional crystallization and column chromatography on silica gel, and the benzoyl protective group is removed, cis-4′-Hydroxyftorafut, trans-3′-hydroxyftorafur, and cis-3′-hydroxyftorafur, which were identified from the PMR spectra and by comparison with the literature data, were obtained.
Similar content being viewed by others
Literature cited
M. Ya. Karpeiskii, S. M. Mikhailov, A. S. Tsieminya, A. A. Ziderman, I. M. Kravchenko, M. Yu. Lidak, and R. A. Zhuk, Khim. Geterotsikl. Soedin., No. 11, 1541 (1980).
S. A. Giller, M. Yu. Lidak, and R. A. Zhuk, Dokl. Akad. Nauk SSSR, 176, 332 (1967).
N. G. Blokhina, N. I. Karev, V. D. Sokolova, and A. M. Lipatov, in: Experimental and Clinical Pharmacotherapy [in Russian], Vol. 7, Zinatne, Riga (1977), p. 208.
I. A. Benvenuto, K. Lu, S. W. Hall, R. S. Benjamin, and T. L. Loo, Cancer Res., 38, 3867 (1978).
I. A. Benvenuto, J. G. Liehr, T. Winkler, D. Farquhar, R. M. Caprioli, and T. L. Loo, Cancer Res., 39, 3199 (1979).
J. L. Au, A. T. Wu, M. A. Friedman, and W. Sadee, Cancer Treat. Rep., 83, 343 (1979).
A. J. Lin, R. S. Benjamin, P. N. Rao, and T. L. Loo, J. Med. Chem., 22, 1096 (1979).
R. B. Meyer and C. H. Levenson, Biochem. Pharmacol., 29, 665 (1980).
C. C. Prise and I. V. Krishnamurti, J. Am. Chem. Soc., 52, 5335 (1950).
Author information
Authors and Affiliations
Additional information
See [1] for Communication 14.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 8, pp. 1094–1096, August, 1981.
Rights and permissions
About this article
Cite this article
Kaulinya, L.T., Zhuk, R.A. & Lidak, M.Y. Analogs of pyrimidine nucleosides. 15. Synthesis of 3′-hydroxy- and 4′-hydroxyftorafur. Chem Heterocycl Compd 17, 813–815 (1981). https://doi.org/10.1007/BF00503665
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00503665