Abstract
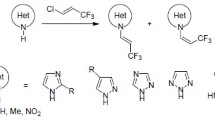
The aminomethylation of oxindole, 1-phenyl-3-methyl-5-pyrazolone, and N-phenyl-rhodanine was studied. Derivatives of these CH acids were obtained as a result of aminomethylation. The addition products were subjected to acid and base hydrolysis; the corresponding arylidene derivatives are formed in the case of the products of aminomethylation of oxindole and 1-phenyl-3-methyl-5-pyrazolone, while thioglycolic acids are formed in the case of N-phenylrhodanine derivatives.
Similar content being viewed by others
Literature cited
N. S. Kozlov and G. V. Vorob'eva, Vestsi Akad. Nauk Belorussk. SSR, No. 4, 107 (1968).
N. S. Kozlov and Z. Z. Nugumanov, Tr. Permsk. Sel'skokhoz. Inst., 38, 38 (1966).
L. S. Povarov, V. I. Grisov, R. A. Karakhanov, and B. M. Mikhailov, Izv. Akad. Nauk SSSR, Ser. Khim., No. 1, 179 (1964).
N. S. Kozlov and Z. A. Abramova, Zh. Obshch. Khim., 32, 2426 (1962).
H. Jenkins and U. Hartung, The Chemistry of Organic Medicinal Preparations [Russian translation], Inostr. Lit., Moscow (1949), p. 592.
I. M. Desmarchelier and R. B. Johns, Org. Mass Spectrom., 2, 697 (1969).
E. Larsen, J. H. Quereshi, and J. Mölier, Org. Mass Spectrom., 7, 89 (1973).
I. S. Shpileva, N. A. Klyuev, F. L. Kolodkin, R. A. Khmel'nitskii, and I. I. Levkoev, Dokl. Akad. Nauk SSSR, 217, 365 (1974).
A. A. Deikalo, Master's Dissertation, Donetsk (1971).
O. N. Chupakhin, V. E. Kirichenko, and I. Ya. Postovskii, Khim. Geterotsikl. Soedin., No. 8, 1116 (1974).
A. N. Rozenberg, G. N. Bogdanov, and A. K. Sheinkman, Khim. Geterotsikl. Soedin., No. 8, 1091 (1972).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 8, pp. 1088–1093, August, 1981.
Rights and permissions
About this article
Cite this article
Pavlenko, N.I., Marshtupa, V.P., Klyuev, N.A. et al. Addition of heterocyclic CH acids to the C=N bond of azomethines. Chem Heterocycl Compd 17, 808–812 (1981). https://doi.org/10.1007/BF00503664
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00503664