Abstract
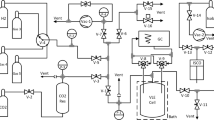
The fugacity coefficients of hydrogen in binary mixtures with ethane were measured. Data were taken using an experimental chamber which is divided into two regions by a semipermeable membrane through which hydrogen, but not ethane, can penetrate. The measurement of the gas pressures inside and outside the membrane gives the hydrogen component fugacity at a given temperature, binary mixture mole fraction, and mixture pressure. In this paper, results are reported at mixture pressures of 5.25, 6.97, 10.21, and 13.47 MPa. In each case, the temperature of the mixture was maintained at an average value of 130°C (403.15 K). The general qualitative features of the data are discussed, and comparisons are made with predictions obtained from the Redlich-Kwong and Peng-Robinson equations of state.
Similar content being viewed by others
References
T. J. Bruno, J. Res. Natl. Bur. Stand. (U.S.) 90(2):127 (1985).
T. J. Bruno, G. L. Hume, and J. F. Ely, Int. J. Thermophys. 7:1033 (1986).
T. J. Bruno and G. L. Hume, Int. J. Thermophys. 7:1053 (1986).
T. J. Bruno, Int. J. Thermophys. 8:205 (1987).
T. J. Bruno and J. A. Schroeder, Int. J. Thermophys. 8:437 (1987).
T. J. Bruno and J. A. Schroeder, Int. J. Thermophys. 9:525 (1988).
T. J. Bruno and S. L. Outcalt, Int. J. Thermophys., in press.
H. Y. Cheh, Proc. 6th Symp. Thermophys. Prop. (ASME, New York, 1963), p. 256.
T. J. Bruno, J. Chromatogr. Sci. 23:325 (1985).
T. J. Bruno and J. G. Shepherd, Anal. Chem. 58:671 (1986).
T. J. Bruno and P. D. N. Svoronos, Basic Tables for Chemical Analysis, National Bureau of Standards (U.S.) Technical Note 1096 (1986).
T. J. Bruno and P. D. N. Svoronos, Handbook of Basic Tables for Chemical Analysis (CRC Press, Boca Raton, Fla., 1988), in press.
T. J. Bruno, Anal. Chem. 58:1596 (1986).
J. F. Ely, Proc. 63rd. Gas Process. Assoc. Com. (1984), p. 9.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bruno, T.J., Schroeder, J.A. & Outcalt, S.L. Hydrogen component fugacity coefficients in binary mixtures with ethane: Pressure dependence. Int J Thermophys 11, 889–896 (1990). https://doi.org/10.1007/BF00503581
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00503581