Summary
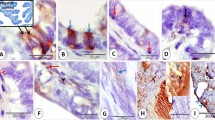
Lectin binding patterns in normal human endometrium were examined by light and electron microscopy using seven different lectins (ConA, WGA, RCA, PNA, UEA-1, DBA, and SBA). For light microscopic observations, criteria based on the incidence and intensity of cells positive for the lectin staining were adopted to evaluate the different staining patterns of the proliferative and secretory endometria obtained by the avidin-biotin-peroxidase complex (ABC) technique. At the light microscopic level, ConA, WGA, and RCA stained endometrial glandular cells in both phases. The number of PNA-positive cells with the binding sites entirely limited to the apical surface tended to be reduced slightly in the secretory phase. UEA-1 weakly stained the apical surface of glandular cells in the proliferative phase but not in the secretory phase. Among the lectins used in this study, DBA and SBA displayed remarkable changes between the phases. That is, in the proliferative phase they produced only a faint or slight positive stain at the apical surface, but the incidence and intensity of DBA- and SBA-positive glandular cells increased in the secretory phase. By electron microscopy, the reaction product of ConA was observed in the plasma membrane, endplasmic reticulum, nuclear envelope, and the Golgi apparatus, and the binding sites of RCA and DBA were observed in the plasma and, Golgi membranes. Between both phases, the reactivity of ConA and RCA showed almost no change. However, the secretory endometrial cells containing the DBA-positive Golgi apparatus were markedly increased in number compared with the proliferative ones bearing the lectin-positive organelles. These results indicate that DBA and SBA are useful for detection of hormone-mediated changes of normal human endometrium, and suggest that such changes are closely related to the Golgi apparatus.
Similar content being viewed by others
References
Aubery M, Guillouzo A, Bernard B, Font J (1980) Changes in Ricinus communis lectin binding to the cell surface of human liver cells in culture. Exp Cell Res 129:273–280
Bernhard W, Avrameas S (1971) Ultrastructural visualization of cellular carbohydrate components by means of concanavalin A. Exp Cell Res 64:232–236
Boland CR, Montogomery CK, Kim YS (1982) A cancer-associated mucin alteration in benign colonic polyps. Gastroenterology 82:664–672
Cavazos F, Green JR, Hall DG, Lucas FV (1967) Ultrastructure of the human endometrial glandular cell during the menstrual cycle. Am J Obstet Gynecol 99:833–854
Ferenczy A, Bertrand G, Gelfand MM (1982) Proliferation kinetics of human endometrium during the normal menstrual cycle. Am J Obstet Gynecol 133:859–867
Goldstein IJ, Hayes CE (1978) The lectins: Carbohydrate-binding proteins of plants and animals. Adv Carbohydr Chem Biochem 35:127–340
Hirano H, Nishiyama F, Numata, H, Hori T (1986a) Cytochemical aspects of glycoconjugates in human anterior pituitary cells. In: Yoshimura F, Gorbman A (eds) Pars distalis of the pituitary gland — Structure, function and regulation. Elsevier, Amsterdam, pp 331–339
Hirano H, Nishiyama F, Yokoyama M (1986b) Regional differences in cytochemical properties of the plasma membrane of rat bladder transitional epithelail cells. In: Hashimoto H, Kuo KH, Lee K, Ogawa K (eds) Recent development of electron microscopy 1985 — Proceedings of the Third Chinese-Japanese Electron Microscopy Seminar. Business Center for Academic Societies Japan, Tokyo, pp 275–284
Hirano H, Parkhous B, Nicolson GL, Lennox ES, Singer SJ (1972) Distribution of saccharide residues on membrane fragments from a myeloma-cell homogenate: its implications for membrane biogenesis. Proc Natl Acad Sci USA 69:2945–2949
Hori T, Nishiyama F, Teramoto A, Matsutani M, Takakura K, Sano K, Hirano H (1982) Lectin-binding sites of the human pituitary adenoma cells by means of the ferritin-labeling technique. Acta Neuropathol 56:67–74
Hori T, Nishiyama K, Teramoto A, Matsutani M, Takakura K, Hirano H (1983) Localization of concanavalin A binding sites in human pituitary adenoma cells as revealed by HRP-labelling method. Acta Neuropathol 62:59–66
Hubbard SC, Ivatt RJ (1981) Synthesis and processing, of asparagine-linked oligosaccharides. Annu Rev Biochem 50:555–583
Ito M, Takata K, Saito S, Aoyagi T, Hirano H (1985) Lectin-binding pattern in normal human gastric mucosa. A light and electron microscopic study. Histochemistry 83:189–193
Kanai Y, Kurohmaru M, Hayashi Y, Nishida T, Kawakami H, Hirano H (1989) Changes in lectin binding pattern of gonads of developing mice. Histochemistry 92:37–42
Klein PJ, Vierbuchen M, Wurz H, Schulz KD, Newman RA (1981) Secretion-associated lectin-binding sites as a parameter of hormone dependence in mammary carcinoma. Br J Cancer 4:746–748
Leathem A, Dokal I, Atkins N (1983) Lectin binding to normal and malignant breast tissue. Diagn Histopathol 6:171–180
Lee MC, Damjanov I (1985) Pregnancy-related changes in the human endometrium revealed by lectin histochemistry. Histochemistry 82:275–280
Lee MC, Wu TC, Wan YJ, Damjanov I (1983) Pregnancy-related changes in the mouse oviduct and uterus revealed by differential binding of fluoresceinated lectins. Histochemistry 79:365–375
Martikainen PM, Makela SI, Santti, RS, Harkonen PL, Suominen JJ (1987) Interaction of male and female sex hormones in cultured rat prostate. Prostate 11:291–303
Nelson JD, Jato-Rodriguez JJ, Labrie F, Mookerjea S (1977) Glycosyltransferase and UDP-galactose pyrophosphatase activities in the endometrium during the oestrous cycle of the rat. J Endocrinol 73:53–58
Nogami H, Nabeya K, Ito M, Yamaguchi Y, Hirano H (1988) Changes in lectin binding pattern of human esophagus in association with malignancy. In: Siewert JR, Hölscher AH (eds) Diseases of the esophagus. Springer, Berlin Heidelberg New York, pp 55–59
Ookusa Y, Takata K, Nagashima M, Hirano H (1983) Distribution of glycoconjugates in normal human skin using biotinyl lectins and avidin-horseradish peroxidase. Histochemistry 79:1–7
Ookusa, Y, Takata K, Nagashima M, Hirano H (1985) Lectinbinding pattern in extramammary Paget's disease by horseradish peroxidase (HRP)-labeling method — Specific staining with Dolichos biflorus agglutinin (DBA). Arch Dermatol Res 277:65–70
Rothman JE (1985) The compartmental organization of the Golgi apparatus. Sci Am 253 (3):84–95
Sugimori H, Ookuma Y, Iwasaki T, Kidera Y (1986) Lectin histochemistry of the cervical malignancies. Acta Obstet Gynecol Jpn 38:111–112
Takata K, Hirano H (1983) Changes in soybean agglutinin (SBA) and peanut agglutinin (PNA) binding pattern in the epidermis of the developing chick embryo. Dev Growth Differ 25:299–305
Wynn RM (1977) Histology and ultrastructure of the human endometrium. In: Wynn RM (ed) Biology of the uterus. Plenum Press, New York, pp 341–376
Yokoyama M, Nishiyama F, Kawai N, Hirano H (1980) The staining of Golgi membrane with Ricinus communis agglutinin-horseradish peroxidase conjugate in mouse tissue cells. Exp Cell Res 125:47–53
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Aoki, D., Kawakami, H., Nozawa, S. et al. Differences in lectin binding patterns of normal human endometrium between proliferative and secretory phases. Histochemistry 92, 177–184 (1989). https://doi.org/10.1007/BF00500916
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00500916