Summary
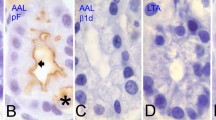
Histochemical analyses of the chemical structures of sugar sequences with or without blood group specificity were carried out by combined stepwise digestion of tissue sections with exo-and endoglycosidases and subsequent lectin stainings in formalin-fixed, paraffin-embedded human pancreas. In acinar cells from blood group A or AB secretor individuals, sequential digestion with α-N-acetylgalactosaminidase and α-L-fucosidase imparted reactivity with peanut agglutinin (PNA) in cells reactive with Dolichos biflorus agglutinin as well as those with Ulex europaeus agglutinin I(UEA-I). Simple fucosidase digestion imparted the PNA reactivity only in UEA-I reactive cells. Sequential digestion with α-galactosidase and fucosidase likewise liberated the PNA binding sites in Griffonia simplicifolia agglutinin I-B4 reactive cells from blood group B and AB secretors. Sialidase digestion liberated the PNA binding sites not only in acinar cells but also intercalated duct cells, islet cells of Langerhans and endothelial cells. The PNA reactivity obtained by these enzyme digestions was eliminted by endo-α-N-acetylgalactosaminidase (endo-GalNAcdase) digestion. Preexisting PNA affinity in acinar cells from nonsecretors was also susceptible to endo-GalNAcdase treatment. Following the endo-GalNAcdase digestion, fucosidase or sialidase digestion recovered the PNA reactivity in acinar cells from nonsecretors. These results show that ABH determinants carried on O-glycosidically linked type 3 chain (D-galactose-(β1-3)-N-acetyl-D-galactosamineα1-serine or threonine) are secreted in pancreatic acinar cells and suggest that product coded by the secretor gene is required for the complete conversion of type 3 precursor chains into H determinants.
Similar content being viewed by others
References
Betteridge A, Watkins WM (1985) Variant forms of α-L-fucosyltransferase in human submaxillary glands from blood group ABH “secretor” and “non-secretor” individuals. Glycoconjugate J 2:61–78
Breimer ME (1984) Tissue specificity of glycosphingolipids as expressed in pancreas and small intestine of blood group A and B human individuals. Arch Biochem Biophys 228:71–85
Donald ASR (1981) A-active trisaccharides isolated from A1 and A2 blood-group-specific glycoprotein. Eur J Biochem 120:243–249
Ebisu S, Iyer S, Goldstein IJ (1987) Equilibrium dialysis and carbohydrate-binding studies on the 2-acetimido-2-deoxy-D-glucopyranosyl-binding lectin from Bandeiraea simplicifolia seeds. Carbohydr Res 61:129–138
Endo Y, Kobata A (1976) Partial purification and characterization of an endo-α-N-acetylgalactosaminidase from the culture medium of Diplococcus pneumoniae. J Biochem 80:1–8
Goldstein IJ, Hayes CE (1978) The lectins Carbohydrate-binding proteins of plants and animals. Adv Carbohydr Chem Biochem 35:127–340
Hanisch F-G, Egge H, Peter-Katalinic J, Uhlenbruck G (1986) Structure of neutral oligosaccharides derived from mucous glycoproteins of human seminal plasma. Eur J Biochem 155:239–247
Ito N, Nishi K, Nakajima M, Matsuda Y, Ishitani A, Mizumoto J, Hirota T (1986) Localization of blood group antigens in human pancreas with lectin-horseradish peroxidase conjugates. Acta Histochem Cytochem 19:205–218
Ito N, Nishi K, Nakajima M, Ishitani A, Okamura Y, Matsuda Y, Hirota T (1987) Histochemical reactivity of soybean agglutinin with blood group antigens and their precursor substances in acinar cells of human pancreas. J Histochem Cytochem 35:881–890
Ito N, Nishi K, Nakajima M, Okamura Y, Hirota T (1988a) Effects of α-L-fucosidase digestion on lectin staining in human pancreas. J Histochem Cytochem 36:503–509
Ito N, Nishi K, Nakajima M, Okamura Y, Hirota T (1988b) Effects of α-galactosidase digestion on lectin staining in human pancreas. Histochemistry 89:121–128
Kumazaki T, Yoshida A (1985) Biochemical evidence that secretor gene, Se, is a structural gene encoding a specific fucosyltransferase. Proc Natl Acad Sci USA 81:4193–4197
Lotan R, Skutelsky E, Danon D, Sharon N (1975) The purification, composition, and specificity of the anti T lectin from peanut (Arachis hypogaea). J Biol Chem 250:8518–8523
Macartney JC (1986) Lectin histochemistry of galactose and N-acetyl-galactosamine glycoconjugates in normal gastric mucosa and gastric cancer and relationship with ABO and secretor status. J Pathol 150:135–144
Nakayama J, Ota M, Honda Y, Katsuyama T (1987) Histochemical demonstration of sugar residues by lectin and immunocytochemical techniques for blood group antigens in human colon. Histochem J 19:454–464
Oriol R, Danilovs J, Hawkins BR (1981) A new genetic model proposing that the Se gene is a structural gene closely linked to the H gene. Am J Hum Genet 33:421–431
Pendu JL, Oriol R, Juszczak G, Liberge G, Rougeer P, Salmon C, Cartron JP (1983) α-L-Fucosyltransferase activity in sera of individuals with H-deficient red cells and normal H antigen in secretions. Vox Sang 44:360–365
Picard JK, Feizi T (1983) Peanut lectin and anti T antibodies reveal structural differences among human gastrointestinal glycoproteins. Mol Immunol 20:1215–1220
Pour PM, Tempero MM, Takasaki H, Uchida E, Takiyama Y, Burnet DA, Steplewski Z (1988) Expression of blood grouprelated antigens ABH, Lewis A, Lewis B, Lewis X, Lewis Y, and CA 19-9 in pancreatic cancer cells in comparison with the patient's blood group type. Cancer Res 48:5422–5426
Rouger PH, Goossens D, Gene P, Salmon CH (1981) Distribution of blood group antigens in adult pancreas. Tissue Antigens 18:51–55
Szulman AE (1960) The histological distribution of blood group substances A and B in man. J Exp Med 111:785–80
Umemoto J, Bhavanandan VP, Davidson EA (1977) Purification and properties of an endo-α-N-acetylgalactosaminidase from Diplococcus pneumoniae. J Biol Chem 252:8609–8614
Watkins WM (1966) Blood-group substances. In the ABO system the genes control the arrangement of sugar residues that determines blood-group specificity. Science 152:172–181
Wherrett JR, Hakomori S-I (1973) Characterization of a blood group B glycolipid, accumulating in the pancreas of a patient with Fabry's disease. J Biol Chem 248:3046–3051
Wood C, Kabat EA, Murphy LA, Goldstein IJ (1979) Immunochemical studies of the combining sites of two isolectins, A4 and B4, isolated from Bandeiraea simplicifolia Arch Biochem Biophys 198:1–11
Yuan M, Itzkowiz SH, Boland CR, Kim YD, Tomita JT, Palekar A, Bennington JL, Trump BF, Kim YS (1986) Comparison of T antigen expression in normal, premalignant, and malignant human colonic tissue using lectin and antibody immunohistochemistry. Cancer Res 46:4841–4847
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ito, N., Nishi, K., Nakajima, M. et al. Histochemical demonstration of O-glycosidically linked, type 3 based ABH antigens in human pancreas using lectin staining and glycosidase digestion procedures. Histochemistry 92, 307–312 (1989). https://doi.org/10.1007/BF00500545
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00500545