Abstract
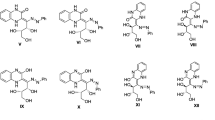
The 1H and 13C NMR spectra of the E and Z isomers of 2-, 3-, and 4-benzoylpyridine oximes and their ethers were analyzed thoroughly, and the 1H-13C spin-spin coupling constants (SSCC) were determined. It was established that the magnitude of the γ effect for the quaternary carbon atoms in the E and Z isomers depends on the site of substitution in the pyridine ring. It was assumed that the intermolecular hydrogen bond is stronger in the E form than in the Z form. The existence of the Z isomer of 2-benzoylpyridine oxime in deuterochloroform with an intramolecular hydrogen bond was proved.
Similar content being viewed by others
Literature Cited
G. W. Buchanan and B. A. Dawson, Can. J. Chem., 55, 1437 (1977).
L. B. Krivdin, V. V. Shcherbakov, and G. A. Kalabin, Zh. Obshch. Khim., 22, 342 (1986).
Yu. Yu. Popelis, é. é. Liepin'sh, é. Ya. Lukevits, Khim. Geterotsikl. Soedin., No. 9, 1172 (1985).
O. M. Glozman, L. A. Zhmurenko, L. M. Meshcheryakov, L. N. Borisova, V. P. Lezina, S. G. Rozenberg, N. A. Novikov, L. N. Nerobkova, A. N. Aliev, Sh. I. Ismailov, T. A. Voronina, and V. A. Zagorevskii, Khim.-farm. Zh., No. 9, 1059 (1987).
A. Yu. Denisov, V. I. Mamatyuk, and O. P. Shkurko, Khim. Geterotsikl. Soedin., No. 9, 1223 (1984).
J. H. Gove, J. Org. Chem., 38, 3517 (1973).
V. I. Ionin, B. A. Ershov, and A. I. Kol'tsov, NMR Spectroscopy in Organic Chemistry [in Russian], Khimiya, Leningrad (1983), p. 167.
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 2, pp. 203–208, February, 1990.
Rights and permissions
About this article
Cite this article
Lezina, V.P., Rozenberg, S.G., Glozman, O.M. et al. Study of three-dimensional structures of benzoylpyridine oximes and their ethers by 1H and 13C NMR spectroscopy. Chem Heterocycl Compd 26, 172–178 (1990). https://doi.org/10.1007/BF00499411
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00499411