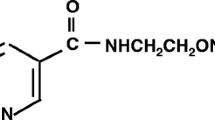
Summary
The influence of a low concentration of 3-isobutyl-1-methylxanthine (IBMX; 5μmol l−1) on the positive inotropic effect of vanadate (NH4VO3; 10–1,000 μmol l−1) and isoprenaline (ISO; 0.1–300 nmol l−1) was studied in electrically driven (frequency 1 Hz) papillary muscles isolated from guinea pigs. Furthermore the influence of maximally effective concentrations of IBMX (100μmol l−1), ISO (100nmol l−1) and dihydro-ouabain (DHO; 70μmol l−1) on the inotropic effect of NH4VO3 was investigated.
-
1.
IBMX (5μmol l−1) shifted the concentrationresponse curve for ISO to the left (EC50-ratio 2.4). In contrast, the positive inotropic effect of NH4VO3 (mean EC50 81.7μmol l−1) was not influenced by IBMX at this concentration.
-
2.
In the presence of high concentrations of IBMX or ISO, NH4VO3 exerted a concentration-dependent (10–1,000μmol l−1) atropine-insensitive negative inotropic effect with mean EC50 values of 40.1 μmol l−1 and 73.0μmol l−1, respectively.
-
3.
In the presence of the maximally effective concentration of DHO, NH4VO3 had no effect on force of contraction, i.e. it neither further increased nor reduced the DHO-induced positive inotropic effect.
-
4.
The negative inotropic effect of NH4VO3 in the presence of IBMX or ISO in guinea-pig papillary muscles was not accompanied by a shortening of the action potential.
-
5.
From the failure of IBMX (5μmol l−1) to enhance the positive inotropic effect of NH4VO3 it is concluded that the recently described small increase in c-AMP, which accompanies the NH4VO3-produced increase in force of contraction in intact papillary muscles, is not of major importance in mediating the agent's positive inotropic effect.
-
6.
The mechanism of the negative inotropic effect of NH4VO3 in the presence of high concentrations of IBMX or ISO remains obscure. Apparently, it is not due to a shortening of the action potential. A stimulation of muscarinic cholinergic receptors is also unlikely although there are obvious similarities between the negative inotropic effect of NH4VO3 and that of acetylcholine described under similar conditions in ventricular heart muscle preparations. Interestingly, all drugs hitherto found capable of evoking a negative inotropic effect of vanadate in ventricular tissue are well known to increase myocardial c-AMP levels.
Similar content being viewed by others
References
Akera T, Takeda K, Yamamoto S, Brody TM (1979) Effects of vanadate on Na+, K+-ATPase and on the force of contraction in guinea-pig hearts. Life Sci 25:1803–1812
Bauer AC, Schwabe U (1980) An improved assay of cyclic 3′, 5′-nucleotide phosphodiesterases with QAE-Sephadex columns. Naunyn-Schmiedeberg's Arch Pharmacol 311:193–198
Beaugé LA, Glynn IM (1977) A modifier of (Na++K+)ATPase in commercial ATP. Nature 268:355–356
Beaugé LA, Glynn IM (1978) Comercial ATP containing traces of vanadate alters the response of (Na++K+)-ATPase to external potassium. Nature 272:551–552
Beavo JA, Rogers NL, Crofford OR, Hardman JG, Sutherland EW, Newman EV (1970) Effects of xanthine derivatives on lipolysis and on adenosine 3′,5′-monophosphodiesterase activity. Mol Pharmacol 6:597–603
Beeler GW Jr, Reuter H (1970) Voltage clamp experiments on ventricular myocardium fibres. J Physiol (Lond) 207:165–190
Bello-Reuss EN, Grady TP, Mazumdar DC (1979) Serum vanadium levels in chronic renal disease. Ann Intern Med 91:743
Borchard U, Fox AAL, Greeff K, Schlieper P (1979) Influence and positive inotropic action of vanadate on atrial and ventricular myocardium. Nature 279:339–341
Borchard U, Fox AAL, Greeff K, Schlieper P (1980) Influence of vanadate on electrophysiological and contractile parameters of atrial myocardium. Basic Res Cardiol 75:423–427
Brown BS, Polson JB, Krzanowski JJ, Wiggins JR (1980) Influence of isoproterenol and methylisobutylxanthine on the contractile and cyclic nucleotide effects of methacholine in isolated rat atria. J Pharmacol Exp Ther 212:325–332
Cantley LC Jr, Josephson L, Warner R, Yanagisawa M, Lechene C, Guidotti G (1977) Vanadate is a potent (Na, K)-ATPase inhibitor found in ATP derived from muscle. J Biol Chem 252:7421–7423
Cantley LC Jr, Cantley LG, Josephson L (1978a) A characterization of vanadate interactions with the (Na, K)-ATPase. Mechanistic and regulatory implications. J Biol Chem 253:7361–7368
Cantley LC Jr, Resh MD, Guidotti G (1978b) Vanadate inhibits the red cell (Na+, K+)ATPase from the cytoplasmic side. Nature 272:552–554
Chamales MH, Gourley RD, Williams BJ (1975) Effect of acetylcholine on changes in contractility, heart rate and phosphorylase activity produced by isoprenaline, salbutamol and aminophylline in the perfused guinea-pig heart. Br J Pharmacol 53:531–538
Charney AN, Silva P, Epstein FH (1975) An in vitro inhibitor of Na-K-ATPase present in an adenosine-triphosphate preparation. J Appl Physiol 39:156–158
Endoh M (1980) The time course of changes in cyclic nucleotide levels during cholinergic inhibition of positive inotropic actions of isoprenaline and theophylline in the isolated canine ventricular myocardium. Naunyn-Schmiedeberg's Arch Pharmacol 312:175–182
Erdmann E (1980) Cardiac effects of vanadate. Basic Res Cardiol 75:411–412
Erdmann E, Krawietz W, Philipp G, Hackbarth I, Schmitz W, Scholz H (1979) Stimulatory effect of vanadate on (Na++K+)-ATPase activity and on 3H-ouabain-binding in a cat heart cell membrane preparation. Nature 278:459–461
Grupp G, Grupp I, Johnson CL, Wallick ET, Schwartz A (1979) Effects of vanadate on cardiac contraction and adenylate cyclase. Biochem Biophys Res Commun 88:440–447
Hackbarth I, Schmitz W, Scholz H, Erdmann E, Krawietz W, Philipp G (1978) Positive inotropism of vanadate in cat papillary muscle. Nature 275:67
Hackbarth I, Schmitz W, Scholz H, Wetzel E, Erdmann E, Krawietz W, Philipp G (1980) Stimulatory effect of vanadate on cyclic AMP levels in cat papillary muscle. Biochem Pharmacol 29:1429–1432
Hopkins LL Jr, Mohr HE (1974) Vanadium as an essential nutrient. Fed Proc 33:1773–1775
Hudgins PM, Bond GH (1979) Reversal of vanadate inhibition of NaK-ATPase by catecholamines. Res Commun Pathol Pharmacol 23:313–326
Inui J, Schümann HJ (1979) Different mode of action of acetylcholine (ACH) on the positive inotropic effect caused by α- or β-adrenoceptor stimulation in the rabbit heart. Naunyn-Schmiedeberg's Arch Pharmacol 308:R33
Korth M (1978) Effects of several phosphodiesterase-inhibitors on guinea-pig myocardium. Naunyn-Schmiedeberg's Arch Pharmacol 302:77–86
Krawietz W, Werdan K, Erdmann E (1979) Stimulatory effect of vanadate on the adenylate cyclase of cardiac tissue. Biochem Pharmacol 28:2517–2520
Krawietz W, Werdan K, Erdmann E (1980) Stimulation of human cardiac adenylate cyclase by vanadate. Basic Res Cardiol 75:433–437
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with Folin phenol reagent. J Biol Chem 193:265–275
McAfee DA, Whiting GJ, Siegel B (1978) Neurotransmitter and cyclic nucleotide modulation of frog cardiac contractility. J Mol Cell Cardiol 10:705–716
McNeill JH, Brenner MJ, Muschek LD (1973) Interaction of four methylxanthine compounds and norepinephrine on cardiac phosphorylase activation and cardiac contractility. Recent Adv Stud Cardiac Struct Metab 3:261–273
Meester WD, Hardman HF (1967) Blockade of the positive inotropic actions of epinephrine and theophylline by acetylcholine. J Pharmacol Exp Ther 158:241–247
Meinertz T, Nawrath H, Scholz H (1976) Possible role of cyclic AMP in the relaxation process of mammalian heart: effects of dibutyryl cyclic AMP and theophylline on potassium contractures in cat papillary muscles. Naunyn-Schmiedeberg's Arch Pharmacol 293:129–137
Nawrath H, Zong X-G (1980) Evidence that cyclic GMP mediates the effects of acetylcholine on mammalian ventricular heart muscle. Naunyn-Schmiedeberg's Arch Pharmacol 311:R36
Nechay BR, Saunders JP (1978) Inhibition by vanadium of sodium and potassium dependent adenosinetriphosphatase derived from animal and human tissues. J Environ Pathol Toxicol 2:247–262
Peytreman A, Nicholson WE, Liddle GW, Hardman JG, Sutherland EW (1973) Effects of methylxanthines on adenosine 3′,5′-monophosphate and corticosterone in the rat adrenal. Endocrinology 92:525–530
Schmitz W, Scholz H, Schumacher A, Erdmann E, Werdan K (1980a) Versuche zur Charakterisierung der positiv inotropen Wirkung von Vanadat. Z Kardiol 69:193
Schmitz W, Hackbarth I, Scholz H, Wetzel E (1980b) Effects of vanadate on the c-AMP system of the heart. Basic Res Cardiol 75:438–443
Scholz H, Hackbarth I, Schmitz W, Wetzel E (1980) Effects of vanadate on myocardial force of contraction. Basic Res Cardiol 75:418–422
Schroeder HA, Balassa JJ, Tipton IH (1963) Abnormal trace metals in man — vanadium. J Chron Dis 16:1047–1071
Schwabe U, Puchstein C, Hannemann H, Söchtig E (1979) Activation of adenylate cyclase by vanadate. Nature 277:143–145
Schwarz K, Milne DB (1971) Growth effects of vanadium in the rat. Science 174:426–428
Simons TJB (1979) Vanadate — a new tool for biologists. Nature 281:337–338
Solaro RJ, Holroyde MJ, Wang T, Matlib MA, Grupp I, Grupp G, Schwartz A (1980) Effects of vanadate on biochemical and contractile properties of rabbit hearts. J Cardiovasc Pharmacol 2:445–452
Sutherland EW, Robinson GA, Butcher RW (1968) Some aspects of the biological role of 3′, 5′-monophosphate (cyclic AMP). Circulation 37:279–306
Terasaki WL, Brooker G (1977) Cardiac adenosine 3′:5′-monophosphate. Free and bound forms in the isolated rat atrium. J Biol Chem 252:1041–1050
Watanabe AM, Besch HR Jr (1975) Interaction between cyclic adenosine monophosphate and cyclic guanosine monophosphate in guinea pig ventricular myocardium. Circ Res 37:309–317
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brückner, R., Schmitz, W., Scholz, H. et al. Negative inotropic effect of vanadate in ventricular myocardium in the presence of 3-isobutyl-1-methylxanthine or isoprenaline. Naunyn-Schmiedeberg's Arch. Pharmacol. 315, 147–153 (1980). https://doi.org/10.1007/BF00499257
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00499257