Summary
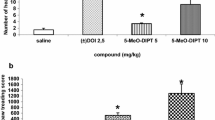
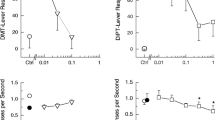
The effects of various tetrahydro-β-carboline (THβC) compounds on serotonin (5-hydroxytryptamine, 5-HT) and 5-hydroxyindoleacetic acid (5-HIAA) concentrations and serotonin synthesis in mouse brain were investigated. 6-Methoxy-THβC (6-MeO-THβC) (100 mg/kg i.p.) significantly increased 5-HT concentrations from 15 min until 4 h after administration, and decreased 5-HIAA concentrations from 15 min until 10 h. The lowest dose of 6-MeO-THβC which produced a significant 5-HT increase was 25 mg/kg whereas 12.5 mg/kg produced a significant decrease of 5-HIAA. Comparisons among the THβC's in their ability to decrease 5-HIAA concentrations showed the following order of potency: THβC>6-MeO-THβC>6-MeO-tetrahydroharman>6-OH-THβC, In order to determine the mechanism for the increase in 5-HT and decrease in 5-HIAA produced by 6-MeO-THβC, its effect was compared to that of reference drugs known to inhibit monoamine oxidase (e. g. clorgyline, deprenyl, pargyline) and synaptosomal 5-HT uptake (chlorimipramine, fluoxetine). These comparisons suggested that the increased 5-HT concentration produced by 6-MeO-THβC was primarily the result of its monoamine oxidase-A inhibiting properties, whereas the decrease in 5-HIAA was the result of a combination of monoamine oxidase-A inhibition and 5-HT uptake inhibition.
Serotonin synthesis in mouse brain following treatment with various tetrahydro-β-carboline compounds and other drugs affecting serotonin metabolism was determined using the so-called in vivo tryptophan hydroxylase method. This involved administering either saline or test drug followed at various times later by an injection of the decarboxylase inhibitor RO4-4602 (800 mg/kg), killing the mouse 30 min later, and measuring 5-hydroxytryptophan (5-HTP) in brain. 6-MeO-THβC at 100 mg/kg decreased the formation of 5-HTP with respect to saline controls from 1 to 4 h after administration, and this decrease was shown to be dose-dependent as measured at 2 h. The other THβC's including THβC, 6-OH-THβC and 6-MeO-tetrahydroharman also decreased 5-HTP formation at 2 h. In order to determine the mechanism for this decreased 5-HTP formation produced by the THβC's, their effects were compared to those of reference drugs known to inhibit monoamine oxidase-A (clorgyline, Lilly 51641), to inhibit synaptosomal serotonin uptake (fluoxetine, chlorimipramine), or to be a serotonin receptor agonist (quipazine). These comparisons suggested that the decrease in 5-HTP formation produced by the THβC's was most likely the result of a combination of monoamine oxidase inhibition plus serotonin uptake inhibition with the latter effect somewhat more potent.
Similar content being viewed by others
References
Atack CV, Magnusson T (1970) Individual elution of noradrenaline (together with adrenaline), dopamine, 5-hydroxytryptamine and histamine from a single, strong cation exchange column, by means of mineral acid-organic solvent mixtures. J Pharm Pharmacol 22:625–627
Atack CV, Magnusson T (1978) A procedure for the isolation of noradrenaline (together with adrenaline), dopamine, 5-hydroxytryptamine and histamine from the same tissue sample using a single column of strongly acidic cation exchange resin. Acta Pharmacol Toxicol 42:35–57
Barker SA, Harrison RE, Brown GB, Christian ST (1979) Gas chromatographic/mass spectrometric evidence for the identification of 1, 2, 3, 4-tetrahydro-β-carboline as an in vivo constituent of rat brain. Biochem Biophys Res Comm 87:146–154
Bedard P, Carlsson A, Lindqvist M (1972) Effects of a transverse cerebral hemisection on 5-hydroxytryptamine metabolism in the rat brain. Naunyn-Schmiedeberg's Arch Pharmacol 272:1–15
Buckholtz NS (1975a) Anticonvulsant effects of 6-methoxy-1, 2, 3, 4-tetrahydro-β-carboline on audiogenic and electroconvulsive seizures in mice. Pharmacol Biochem Behav 3:65–68
Buckholtz NS (1975b) 6-Methoxy-1, 2, 3, 4-tetrahydro-β-carboline produces a retention deficit for one-trial passive avoidance in mice. Behav Biol 14:95–101
Buckholtz NS, Boggan WO (1977a) Monoamine oxidase inhibition in brain and liver produced by β-carbolines: structure-activity relationships and substrate specificity. Biochem Pharmacol 26:1991–1996
Buckholtz NS, Boggan WO (1977b) Inhibition by β-carbolines of monoamine uptake into a synaptosomal preparation: structure-activity relationships. Life Sci 20:2093–2100
Carlsson A, Lindqvist M (1972) The effect of L-tryptophan and some psychotropic drugs on the formation of 5-hydroxytryptophan in the mouse brain in vivo. J Neural Trans 33:23–43
Carlsson A, Lindqvist M (1973) In vivo measurements of tryptophan and tyrosine hydroxylase activities in mouse brain. J Neural Trans 34:79–91
Carlsson A, Lindqvist M (1978) Effects of antidepressant agents on the synthesis of brain monoamines. J Neural Trans 43:73–91
Carlsson A, Kehr W, Lindqvist M (1976) The role of intraneuronal amine levels in the feedback control of dopamine, noradrenaline and 5-hydroxytryptamine synthesis in rat brain. J Neural Trans 39:1–19
Friedman E, Meller E, Hallock M, Afflitto J (in press) Serotonin uptake in rat brain: lack of conformational specificity by potent inhibitory analogues. Mol Pharmacol
Fuentes JA, Garzon J, DelRio J (1977) Potentiation of morphine analgesia in mice after inhibition of brain type B monoamine oxidase. Neuropharmacology 16:857–862
Fuller RW, Perry KW, Molloy BB (1974) Effects of an uptake inhibitor on serotonin metabolism in rat brain: studies with 3-(p-trifluoromethylphenoxy)-N-methyl-3-phenylpropylamine (Lilly 110140). Life Sci 15:1161–1171
Fuller RW, Snoddy HD, Perry KW, Roush BW, Molloy BB, Bymaster FP, Wong DT (1976) The effects of quipazine on serotonin metabolism in rat brain. Life Sciences 18:925–934
Fuller RW, Hemrick SK, Mills J (1978) Inhibition of monoamine oxidase by N-phenacylcyclopropylamine. Biochem Pharmacol 27:2255–2261
Fuxe K, Holmstedt B, Jonsson G (1972) Effects of 5-methoxy-N,N-dimethyl-tryptamine on central monoamine neurons. Eur J Pharmacol 19:25–34
Halaris AE, Lovell RA, Freedman DX (1973) Effect of chlorimipramine on the metabolism of 5-hydroxytryptamine in the rat brain. Biochem Pharmacol 22:2200–2202
Hamon M, Bourgoin S, Enjalbert A, Bockaert J, Hery F, Ternaux JP, Glowinski J (1976) The effects of quipazine on 5-HT metabolism in the rat brain. Naunyn-Schmiedeberg's Arch Pharmacol 294:99–108
Ho BT, McIsaac WM, Walker KE, Estevez V (1968) Inhibitors of monoamine oxidase-influence of methyl substitution on the inhibitory activity of β-carbolines. J Pharm Sci 57:269–274
Ho BT, Taylor D, Walker KE, McIsaac WM (1973) The mode of action of 6-methoxy-1, 2, 3, 4-tetrahydro-β-carboline on brain serotonin. Can J Biochem 51:482–485
Honecker H, Rommelspacher H (1978) Tetrahydronorharmane (tetrahydro-β-carboline), a physiologically occurring compound of indole metabolism. Naunyn-Schmiedeberg's Arch Pharmacol 305:135–141
Jacoby JH, Howd RA, Levin MS, Wurtman RJ (1976) Mechanisms by which quipazine, a putative serotonin receptor agonist, alters brain 5-hydroxyindole metabolism. Neuropharmacology 15: 529–534
Jonsson J, Lewander T (1970) A method for the simultaneous determination of 5-hydroxy-3-indoleacetic acid (5-HIAA) and 5-hydroxy-tryptamine (5-HT) in brain tissue and cerebrospinal fluid. Acta Physiol Scand 78:43–51
Karasawa T, Nakamura J, Masanao S (1974) Simultaneous micro-determination of homovanillic acid and 5-hydroxyindoleacetic acid in brain tissue using Sephadex G-10 and QAE-Sephadex A-25. Life Sci 15:1465–1474
Kellar KJ, Elliott GR, Hollman RB, Vernikos-Danellis J, Barchas JD (1976) Tryptoline inhibition of serotonin uptake in rat forebrain homogenates. J Pharmacol Exp Ther 198:619–625
Lindqvist M (1971) Quantitative estimation of 5-hydroxy-3-indole acetic acid and 5-hydroxytryptophan in the brain following isolation by means of a strong cation exchange column. Acta Pharmacol Toxicol 29:303–313
Maickel RP, Miller FP (1966) Fluorescent products formed by reaction of indole derivatives and O-phthalaldehyde. Anal Chem 38:1937–1938
Marco EJ, Meek JL (1979) The effects of antidepressants on serotonin turnover in discrete regions of rat brain. Naunyn-Schmiedeberg's Arch Pharmacol 306:75–79
McIsaac WM, Taylor D, Walker KE, Ho BT (1972) 6-Methoxy-1,2,3,4-tetrahydro-β-carboline-a serotonin elevator. J Neurochem 19:1203–1206
Meller E, Friedman E, Schweitzer JW, Friedhoff AJ (1977) Tetrahydro-β-carbolines; specific inhibitors of type A monoamine oxidase in rat brain. J Neurochem 28:995–1000
Neff NH, Yang H-YT, Fuentes JA (1974) The use of selective monoamine oxidase inhibitor drugs to modify amine metabolism in brain. Adv Biochem Psychopharmacol 12:49–57
Rommelspacher H, Subramanian N (1979) Tetrahydronorharmane modulates the depolarisation — induced efflux of 5-hydroxytryptamine and dopamine and is released by high potassium concentration from rat brain slices. Eur J Pharmacol 56:81–86
Rommelspacher H, Strauss SM, Rehse K (1978a) β-Carbolines: a tool for investigating structure-activity relationships of the high-affinity uptake of serotonin, noradrenaline, dopamine, GABA and choline into a synaptosome — rich fraction of various regions from rat brain. J Neurochem 30:1573–1578
Rommelspacher H, Strauss S, Cohnitz CH (1978b) Inhibition of 5-hydroxytryptamine uptake by tetrahydronorharmane in vivo. Naunyn-Schmiedeberg's Arch Pharmacol 303:229–233
Rommelspacher H, Honecker H, Barbey M, Meinke B (1979) 6-Hydroxy-tetrahydronorharmane (6-hydroxy-tetrahydro-β-carboline), a new active metabolite of indole-alkylamines in man and rat. Naunyn-Schmiedeberg's Arch Pharmacol 310:35–41
Rosecrans JA, Lovell RA, Freedman DX (1967) Effects of lysergic acid diethylamide on the metabolism of 5-hydroxytryptamine. Biochem Pharmacol 16:2011–2021
Shoemaker DW, Cummins JT, Bidder TG (1978) β-Carbolines in rat arcuate nucleus. Neuroscience 3:233–239
Sparks DL, Buckholtz NS (1978) 6-Methoxy-1,2,3,4-tetrahydro-β-carboline: a specific monoamine oxidase-A inhibitor in CF1 mice. Soc Neurosci Abst 4:434
Sparks DL, Buckholtz NS (1980) Effects of 6-methoxy-1,2,3,4-tetrahydro-β-carboline (6-MeO-THβC) on audiogenic seizures in DBA/2J mice. Pharmacol Biochem Behav 12:119–124
Tachiki KH, Aprison MH (1975) Fluorometric assay for 5-hydroxytryptophan with sensitivity in the picomole range. Anal Chem 47:7–11
Thomas TN, Buckholtz NS, Zemp JW (1979) 6-Methoxy-1,2,3,4-tetrahydro-β-carboline effects on retinal serotonin. Life Sci 25:1435–1442
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Buckholtz, N.S. Brain serotonin and 5-hydroxyindoleacetic acid concentrations and serotonin synthesis following tetrahydro-β-carboline administration in mice. Naunyn-Schmiedeberg's Arch. Pharmacol. 314, 215–221 (1980). https://doi.org/10.1007/BF00498542
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00498542