Summary
Limitations in mass and momentum transfer coupled with high hydrostatic pressures create significant spatial variations in dissolved gas concentrations in large fermenters. Microorganisms are subjected to fluctuating environmental conditions as they pass through the zones in a stirred vessel or along a closed loop fermenter.
A 7-litre fermenter was modified to simulate the dissolved gas and hydrostatic pressure gradients in large vessels.
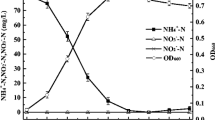
The effect of cycling dissolved oxygen tension (DOT) on penicillin production by Penicillium chrysogenum P1 was investigated. The fermentation was affected by evironmental conditions such as medium composition, pH, size of inoculum, stirrer speed and DOT. Inoculum size below 10% (v/v) and stirrer speeds above 850 rpm caused significant reductions in specific prenicillin production rates (qpen). qpen values were measured at different constant DOT levels. Below 30% air saturation qpen decreased sharply and no production was observed at 10%. Penicillin synthesis was impaired irreversibly below 10% DOT. The same profile was observed at higher stirrer speeds and air flow rates indicating that the effect was a physiological one. Oxygen uptake of the culture was affected significantly below 7% DOT, demonstrating that the critical DOT values for penicillin production and oxygen uptake are two distinct parameters. Carrying out the fermentation at one atmosphere over pressure was found to have no effect. When the dissolved oxygen concentration of the culture medium was cycled around the critical DOT for penicillin production, a considerable decrease in the specific penicillin production rate was observed. The effect was reversible but not transient, indicating a shift in cell metabolism.
These results demonstrate the unfavourable effect of fluctuating environmental conditions on culture performance in stirred tanks. They suggest that these effects should be accounted for during strain selection, process development and scale up stages of an industrial process if the productivities in small scale vessels are to be obtained.
Similar content being viewed by others
References
Anderson RF, Tornqvist EGM (1956) Effect of Oil in Pilot Plant Fermentations for Penicillin Production. J Agr Food Chem 4: 556–559
Bayer PV (1981) Acceleration of Growth in Microbiological Processes. Unpublished report, Giovanola Freres SA, Montley, Schweiz
Bernard A, Cooney CL (1981) Studies on Oxygen Limitations in the Penicillin Fermentation. Eur Cong Biotechnol
Bryant J (1977) The Characterization of Mixing in Fermenters. Adv Biochem Eng 5:101–123
Bundgaard H, Ilver K (1972) A New Spectrophotometric Method for the Determination of Penicillins. J Pharm Pharmacol 24: 790–794
Burton K (1956) A Study of the Conditions and Mechanism of the Diphenylamine Reaction for the Colorimetric Estimation of Deoxyribonucleic Acid. Biochem J 62:315–323
Calam CT (1967) Media for Industrial Fermentations. Process Biochem June: 19–22
Calam CT (1976) Starting Investigational and Production Cultures. Process Biochem Apr: 7–12
Calam CT, Smith GM (1981) Regulation of the Biochemistry and Morphology of Penicillium chrysogenum in Relation to Initial Growth. FEMS Microbiol Lett 10:231–234
Camposano A, Chain EB, Gualandi G (1959) The Effect of Mechanical Agitation on the Production of Kojic Acid in Submerged Culture. Sel Sci Pap Ist Sup Sanita 2:224–240
Charles M (1978) Technical Aspects of the Rheological Properties of Microbial Cultures. Adv Biochem Eng 8:1–62
Evans JI, Hall MJ (1971) Foams and Antifoams in Fermentation. Process Biochem Apr: 23–26
Fewkes RJ (1977) Physical Parameters Govering the Biological Activity of a Non-Newtonian Mycelial Fermentation Broth. PhD Thesis, MIT
Giona AR, Marelli L, Toro L, De Santis R (1976a) Kinetic Analysis of Penicillin Production by Semicontinuous Fermenters. Biotechnol Bioeng 18:473–492
Giona AR, De Santis R, Marelli L, Toro L (1976b) The Influence of Oxygen Concentration and of Specific Rate of Growth on the Kinetics of Penicillin Production. Biotechnol Bioeng 18: 492–512
Gurr E, MacConaill MA (1965) The Rational Use of Dyes in Biology, Leonard Hill Co, London
Gürtler H (1980) Unpublished Report
Harrigan WF, McCane ME (1966) Laboratory Methods in Microbiology, Academic Press, London
Jansen PH, Slott S, Gürtler H (1978) Determination of Mixing Times in Large-Scale Fermenters Using Radioactive Isotopes. Eur Congr Biotechnol I, Part 2, Interlaken, pp 80–82
Johnson MJ (1946) Metabolism of Penicillin-Producing Moulds. Ann NY Acad Sci 48:57–66
Katinger HWD (1976) Physiological Response of Candida tropicalis Grown on n-Paraffin to Mixing in a Tubular Closed Loop Fermenter. Eur J Appl Microbiol Biotechnol 3:103–114
König B, Seewald C, Schügerl K (1981a) Untersuchungen zur Penicillin-Produktion unter verfahrenstechnischen Gesichtspunkten. Chem Ing Technol 53:56–57
König B, Seewald C, Schügerl K (1981b) Process Engineering Investigations of Penicillin Production. Eur J Appl Microbiol Biotechnol 12:205–211
Kubicek CP, Führer L, Zehentrgruber O, Röhr M (1978) The Operation of Oxidative Mechanisms During Citric Acid Fermentation by Aspergillus niger. Eur Congr Biotechnol I, Part 1, Interlaken, pp 131–134
Kubicek, CP, Zehentgruber O, El-Kalak H, Röhr M (1980) Regulation of Citric Acid Production by Oxygen: Effect of Dissolved Oxygen Tension on Adenylate Levels and Respiration in Aspergillus niger. Eur J Appl Microbiol Biotechnol 9:101–115
Lengyel ZL, Nyiri L (1965) The Inhibitory Effect of Carbon Dioxide on Penicillin Biosynthesis. Antibiot Adv Res Prod Clin Use Proc Congr Prague: 733–735
McIntosh AF, Meyrath J (1965) Size of Inoculum and Nitrogen Metabolism in Aspergillus oryzae. J Gen Microbiol 33:57–62
Metz B, Kossen NWF (1977) The Growth of Molds in the Form of Pellets. Biotechnol Bioeng 19:781–799
Metz B, Kossen NWF, van Suijdam JC (1979) The Rheology of Mould Suspensions. Adv Biochem Eng 11:103–156
Meyrath J, McIntosh AF (1965) Size of Inoculum and Carbon Metabolism in Some Aspergillus Species J Gen Microbiol 33: 47–56
Meyrath J, Suchanek G (1972) Inoculation Techniques — Effects due to Quality and Quantity of Inoculum. In: Norris JR, Ribbons DW (eds) Methods in Microbiology, vol 7B. Academic Press, London, pp 159–209
Ojha MN, Meyrath J (1967) Self-Stimulation and Self-Inhibition in Penicillium chrysogenum. Pathol Microbiol 30:959–965
Pan CH, Hepler L, Elander RP (1975) The Effect of Iron on a High Yielding Industrial Strain of Penicillium chrysogenum and Production Levels of Penicillin G. J Ferment Technol 53:854–861
Phillips DH, Johnson MJ (1961) Aeration in Fermentations. J Biochem Microbiol Technol Eng 3:277–309
Pickett AM, Bazin MJ, Topiwala HH (1979) Growth and Composition of Escherichia coli Subjected to Square Wave Perturbations in Nutrient Supply: Effect of Varying Frequencies. Biotechnol Bioeng 21:1043–1055
Placek J, Ujcova E, Musilkova M, Seichert L, Fencl Z (1981) The Role of Mechanical Shear and Cavitations on the Behaviour of Moulds. Inst Microbiol Czech Akad Sci Prague
Rao LK, Mathur DK (1975) Studies on the Production of Bacterial Rennet in a Pilot Plant Fermenter Biotechnol Bioeng 17:1349–1361
Rolinson GN (1952) Respiration of Penicillium chrysogenum in Penicillin Fermentations. J Gen Microbiol 6:336–343
Rolinson GN, Lumb M (1953) The Effect of Aeration on the Utilization of Respiratory Substrates by Penicillium chrysogenum in Submerged Culture. J Gen Microbiol 8:265–272
Ryu DY, Humphrey AE (1972) Reassessment of Oxygen Transfer Rates in Antibiotic Fermentations. J Ferment Technol 50:423–431
Smith MG, Calam CT (1980) Variations in Inocula and Their Influence on the Productivity of Antibiotic Fermentations. Biotechnol Lett 2:261–266
Squires RW (1972) Regulation of the Penicillin Fermentation by means of a Submerged Oxygen-Sensitive Electrode. Dev Ind Microbiol 13:128–135
Steel R (1959) Environmental Control of Antibiotic Synthesis, Some Effects of Agitation on Antibiotic Production in Submerged Fermentation. VIIth Int Bot Congr, Montreal
Tanaka H, Ueda K (1975) Kinetics of My celial Growth Accompanied by Leakage of Intracellular Nucleotides Caused by Agitation. J Ferment Technol 53:27–34
Vardar F (1981) Effect of Cycling Dissolved Oxygen Concentrations on Product Formation in Penicillin Fermentations. PhD Thesis, University College London
Vardar F, Lilly MD (1982) The Measurement of Oxygen Transfer Coefficients on Fermenters by Frequency Response Techniques. Biotechnol Bioeng (in press)
Wang DIC, Fewkes RCL (1977) Effect of Operating and Geometric Parameters on the Behaviour of Non-Newtonian, My celial, Antibiotic Fermentations. Dev Ind Microbiol 18:39–56
Weinberg ED (1970) Biosynthesis of Secondary Metabolites: Roles of Trace Metals. Adv Microbial Physiol 4:1–44
Welles JB, Blanch HW (1976) The Effect of Discontinuous Feeding on Ethanol Production by Saccharomyces cerevisiae. Biotechnol Bioeng 18:129–132
Ziegler H, Dunn IJ, Bourne JR (1978) Mass Transfer and Myceliali Growth in a Tubular Loop Fermenter. Eur Congr Biotechnol I, Part 1, Interlaken: 74–77
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vardar, F., Lilly, M.D. Effect of cycling dissolved oxygen concentrations on product formation in penicillin fermentations. European J. Appl. Microbiol. Biotechnol. 14, 203–211 (1982). https://doi.org/10.1007/BF00498464
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00498464