Abstract
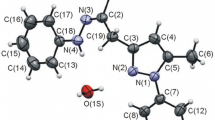
The reaction of N′-methyl-N′-phenylhydrazides of N-acyl-β-phenyl-β-alanine with phosphorus oxychloride is accompanied by cleavage of the C-N bond in the intermediately formed 2-aminoindole derivative and, after splitting out of an amide fragment, leads to 1-methyl-3-benzylidene-2-iminoindoline and nitriles. Pyrimido [4,5-b] indoles — products of cyclization of the corresponding intermediate 2-aminoindole derivatives — were isolated from the reaction mixtures in low yields. The structure of 1-methyl-3-benzylidene-2-iminoindoline was proved by alternative synthesis.
Similar content being viewed by others
Literature cited
A. N. Kost, V. G. Zabrodnyaya, Yu. N. Portnov, and V. G. Voronin, Khim. Geterotsikl. Soedin., No. 4, 484 (1980).
A. N. Kost, R. S. Sagitullin, T. V. Mel'nikova, and G. V. Kaplun, Khim. Geterotsikl. Soedin., No. 10, 1368 (1972).
A. Hantzsch and O. W. Schulze, Ber., 28, 673 (1895).
R. S. Sagitullin, A. N. Kost, E. D. Matveeva, and N. I. Nemudrova, Khim. Geterotsikl. Soedin., No. 7, 920 (1970).
Author information
Authors and Affiliations
Additional information
Deceased.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1356–1359, October, 1980.
Rights and permissions
About this article
Cite this article
Zabrodnyaya, V.G., Portnov, Y.N., Voronin, V.G. et al. Reaction of phenylhydrazides of N-acyl-β-phenyl-β-alanine with phosphorus oxychloride. Chem Heterocycl Compd 16, 1028–1031 (1980). https://doi.org/10.1007/BF00496603
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00496603