Abstract
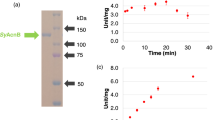
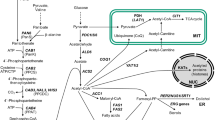
ATP:citrate lyase (EC 4.1.3.8) has been identified in cell-free extracts from the filamentous fungus Aspergillus niger. The enzyme was located in the cytosol. It exhibits an activity at least ten times that of acetate-CoA-kinase (EC 6.2.1.1) during growth on carbohydrates as carbon sources, and is thus considered responsible for acetyl-CoA formation under these conditions. It is formed constitutively and its biosynthesis does not appear to be controlled by changes in the nitrogen or carbon source or type. ATP:citrate-lyase appears to be very labile during conventional purification procedures; a method involving fast protein liquid anion exchange chromatography was thus developed in order to obtain enzyme preparations sufficiently free of enzymes which could interfere with kinetic investigations. This preparation displays commonly known characteristics of ATP:citrate lyase with respect to substrate affinities and cofactor requirements, with the exception that the affinity for citrate is rather low (2.5 mM). No activator was found. The enzyme is inhibited by nucleoside diphosphates, nucleoside monophosphates and palmitoyl-CoA. Regulation of ATP:citrate lyase be the energy charge of the cytosol in relation to lipid or citric acid accumulation is discussed in view of these findings.
Similar content being viewed by others
References
Attwood MM (1973) ATP citrate lyase activity in fungal extracts. Antonie van Leeuwenhoek J Microbiol Serol 39:539–544
Bergmeyer HU, Moellering H (1974) Acetat-Bestimmung mit vorgeschalteter Indicatorreaktion. In: Bergmeyer HU (ed) Methoden der enzymatischen Analyse, vol 2. Verlag Chemie, Weinheim, pp 1566–1574
Boulton CA, Ratledge C (1981) Correlation of lipid accumulation in yeast with possession of ATP-citrate lyase. J Gen Microbiol 127:169–176
Boulton CA, Ratledge C (1983) Partial purification and some properties of ATP-citrate lyase from the oleaginous yeast Lipomyces starkeii. J Gen Microbiol 129:2863–2869
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye-binding. Anal Biochem 72:248–254
Evans CT, Ratledge C (1985) The physiological significance of citric acid in the control of metabolism in lipid-accumulating yeasts. Biotechnol Genet Engin Rev 3:349–375
Habison A, Kubicek CP, Röhr M (1983) Phosphofructokinase from Aspergillus niger: partial purification and regulatory properties. Biochem J 209:669–676
Hoffmann GE, Andres H, Weiss L, Kreisel C, Sander R (1980) Lipogenesis in man: properties and organ distribution of ATP citrate lyase. Biochim Biophys Acta 620:151–158
Kubicek CP, Röhr M (1977) Influence of manganese on enzyme synthesis and citric acid accumulation in Aspergillus niger. Eur J Appl Microbiol 4:167–173
Kubicek CP, Röhr M (1978) The role of the tricarboxylic acid cycle in citric acid accumulation by Aspergillus niger. Eur J Appl Microbiol Biotechnol 5:263–271
Kubicek CP, Röhr M (1986) The citric acid fermentation. CRC Crit Rev Biotechnol 3:331–373
Kubicek CP, Hampel W, Röhr M (1979) Manganese deficiency leads to elevated amino acid pools in citric acid accumulating Aspergillus niger. Arch Microbiol 123:73–79
Mahlen A (1973) Purification and some properties of ATP-citrate lyase from Penicillium spiculisporum. Eur J Biochem 36:342–346
Meixner-Monori B, Kubicek CP, Habison A, Kubicek-Pranz EM, Röhr M (1985) Presence and regulation of the α-ketoglutarate dehydrogenase multienzyme complex from the filamentous fungus Aspergillus niger. J Bacteriol 161:265–271
Meixner-Monori B, Kubicek CP, Harrer W, Schreferl G, Röhr M (1986) NADP-specific isocitrate dehydrogenase from the citric acid accumulating fungus Aspergillus niger. Biochem J 236:549–557
Ng AML, Lloyd GI, Smith JE (1974) Esterase activity of Aspergillus niger in continuous culture. Arch Microbiol 97:209–216
Orthofer R, Kubicek CP, Röhr M (1979) Lipid levels and manganese deficiency in citric acid producing strains of Aspergillus niger. FEMS Microbiol Lett 5:403–406
Osmani SA, Scrutton MC (1983) The subcellular localization of pyruvate carboxylase and of some other enzymes in Aspergillus nidulans. Eur J Biochem 133:551–558
Redshaw JC, Loten EG (1981) A rapid purification of hepatic ATP-citrate lyase using Blue-Sepharose. FEBS Lett 123:261–264
Röhr M, Stadler PJ, Salzbrunn WOJ, Kubicek CP (1979) An improved method for assessing citric acid production by conidia of Aspergillus niger. Biotechnol Lett 1:281–286
Röhr M, Kubicek CP, Kominek J (1983a) Citric acid. In: Reed G, Rehm HJ (eds) Biotechnology, vol 3. Verlag Chemie, Weinheim, pp 420–454
Röhr M, Zehentgruber O, Orthofer R, Kubicek CP (1983b) A balance of carbon and oxygen conversion rates during pilot plant citric acid fermentation by Aspergillus niger: the identification of polyols as major by-products. Int J Microbiol 1:19–25
Shah VK, Ramakrishnan CV (1963) Studies on acid metabolism in Aspergillus niger: part IV: acetyl-CoA-kinase in Aspergillus niger. Enzymologia 26:53–64
Srere PA (1962) Citrate cleavage enzyme. Meth Enzymol 5:641–644
Srere PA (1975) Enzymology of the formation and breakdown of citrate. Adv Enzymol 43:57–101
Wongchai V, Jefferson WE Jr (1974) Pyruvate carboxylase of Aspergillus niger: partial purification and some properties. Fed Proc 33:1378
Zehentgruber O, Kubicek CP, Röhr M (1980) Alternate oxidase from Aspergillus niger. FEMS Microbiol Letts 8:71–74
Author information
Authors and Affiliations
Additional information
Present address: Institut für Allgemeine Biochemie, Universität Wien, Währingerstrasse 38, A-1090 Wien, Austria
Rights and permissions
About this article
Cite this article
Pfitzner, A., Kubicek, C.P. & Röhr, M. Presence and regulation of ATP:citrate lyase from the citric acid producing fungus Aspergillus niger . Arch. Microbiol. 147, 88–91 (1987). https://doi.org/10.1007/BF00492910
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00492910