Abstract
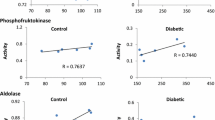
HGPRT from patient M. Y. (enzyme level 0.1% normal) retained a normal apparent K m both for PRPP and for hypoxanthine and was inhibited by its product. The enzyme was, however, unstable at 50 C (43% of activity remaining after 1 hr) when compared with normal controls (81% of activity retained). The enzyme from patient J.D. (enzyme level 0.005% normal) was also unstable (32% of activity retained). Unlike for M.Y., however, all the other characteristics studied were also altered. The enzyme activity was enhanced rather than inhibited by its product (IMP), and the apparent K m (hypoxanthine) could not be calculated due to the sigmoid nature of the curve. Obviously, there is marked heterogeneity in the nature of the biochemical lesion responsible for the Lesch-Nyhan syndrome in these patients, and this is discussed.
Similar content being viewed by others
References
Bakay, B., and Nyhan, W. L. (1972). Electrophoretic properties of hypoxanthine-guanine phosphoribosyl transferase in erythrocytes of subjects with Lesch-Nyhan syndrome. Biochem. Genet. 6139.
Kelley, W. N., and Meade, J. C. (1971). Studies on hypoxanthine-guanine phosphoribosyl-transferase in fibroblasts from patients with the Lesch-Nyhan syndrome: Evidence for genetic heterogeneity. J. Biol. Chem. 2462953.
Kelley, W. N., Rosenbloom, F. M., Henderson, J. F., and Seegmiller, J. E. (1967). A specific enzyme defect in gout associated with overproduction of uric acid. Proc. Natl. Acad. Sci. 571735.
Krenitsky, J. A., Papaioanou, P., and Elion, G. B. (1969). Human hypoxanthine phosphoribosyltransferase. I. Purification, properties and specificity. J. Biol. Chem. 2441263.
Lesch, M., and Nyhan, W. L. (1964). A familial disorder in uric acid metabolism and central nervous system function. Am. J. Med. 36561.
McDonald, J. A., and Kelley, W. N. (1971). Lesch-Nyhan syndrome: Altered kinetic properties of mutant enzyme. Science 171689.
Owen, R. D. (1963). In Burdette, W. J. (ed.), Methodology in Mammalian Genetics, Holden-Day, New York, p. 364.
Rubin, C. S., Dancis, J., Yip, L. C., Nowinski, R. C., and Balis, M. E. (1971). Purification of IMP: pyrophosphate phosphoribosyltransferases. Catalytically incompetent enzymes in Lesch-Nyhan disease. Proc. Natl. Acad. Sci. 681461.
Author information
Authors and Affiliations
Additional information
This research was supported by the Medical Research Council of Canada (a Postdoctoral Fellowship to B.J.R. and Grant No. MA-4061 to J.L.H.) and by the Children's Hospital Research Foundation.
Rights and permissions
About this article
Cite this article
Richardson, B.J., Ryckman, D.L., Komarnicki, L.M. et al. Heterogeneity in the biochemical characteristics of red blood cell hypoxanthine-guanine phosphoribosyl transferase from two unrelated patients with the lesch-nyhan syndrome. Biochem Genet 9, 197–202 (1973). https://doi.org/10.1007/BF00487450
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00487450