Abstract
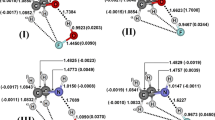
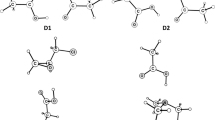
Proton affinity of furazane, furoxane, and their aminoderivatives for protonation at various centers was calculated by ab initio methods using the STO-3G basis set. Basicity was found to decrease in the following order: N-oxide oxygen atom, endocyclic nitrogen atom, amino group. The presence in the ring of aminogroups or N-oxide functions leads to elevated proton affinity of the basic centers which are located in the active positions of the ring.
Similar content being viewed by others
Literature Cited
V. G. Andrianov, M. A. Shokhen, and A. V. Eremeev, Khim. Geterotsikl. Soedin., No. 9, 1221 (1989).
J. Elguero, C. Marzin, A. R. Katritzky, and P. Linda, The Tautomerism of Heterocycles, Academic Press, New York, San Francisco, and London (1976), p. 413.
V. G. Andrianov, M. A. Shokhen, A. V. Eremeev, and S. V. Barmina, Khim. Geterotsikl. Soedin., No. 1, 54 (1987).
D. Viterbo and A. Serafino, Acta Cryst. Sect. B, Struct. Crystallogr. Cryst. Chem., 34, 3444 (1978).
B. T. Storey, W. W. Sullivan, and C. L. Moyer, J. Org. Chem., 29, 3118 (1964).
J. Elguero, E. Gonzalez, and R. Jacquier, Bull. Soc. Chim. Fr., No. 10, 5009 (1968).
I. I. Grandberg, S. V. Tabak, and A. N. Kost, Khim. Geterotsikl. Soedin., No. 1, 85 (1966).
G. Dedichen, Berichte, 39, 1831 (1906).
M. G. Voronkov, T. V. Kamik, V. V. Makarskii, V. A. Lopyrev, S. M. Ponomareva, and E. F. Shibanova, Dokl. Akad. Nauk, 227, 1116 (1976).
I. V. Tselinskii, S. F. Mel'nikova, and S. N. Vergizov, Khim. Geterotsikl, Soedin., No. 3, 321 (1981).
A. F. Pozharskii, Theoretical Principles of Heterocyclic Chemistry [in Russian], Khimiya, Moscow (1985), p. 119.
Author information
Authors and Affiliations
Additional information
For Communication 4, see [1].
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 9, pp. 1261–1264, September, 1989.
Rights and permissions
About this article
Cite this article
Andrianov, V.G., Shokhen, M.A. & Eremeev, A.V. Dependence of the reactivity of five-membered aromatic heterocycles on their structure. 5. Protonation of furazane, furoxane, and their aminoderivatives. Chem Heterocycl Compd 25, 1056–1059 (1989). https://doi.org/10.1007/BF00487310
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00487310