Abstract
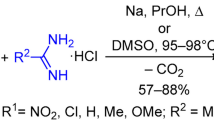
A number of 6-aminophenanthridines have been synthesized; upon alkylation, these compounds form amidinium salts. Depending on the structure of the original amidine, the alkylation may proceed at the exocyclic (n-alkyl), phenanthridine (N-aryl), or pyridine [N-(2-pyridyl)] nitrogen atom.
Similar content being viewed by others
Literature Cited
L. Walls, in: Heterocyclic Compounds, Vol. 4, R. C. Elderfield (ed.), Wiley, New York (1951).
R. J. Grout, in: The Chemistry of Amidines and Imidates, S. Patai (ed.), Interscience, London (1975), p. 255.
V. G. Granik, Usp. Khim., 52, 669 (1983).
A. G. Mikhailovskii, in: Summaries of Papers from 10th Conference of Young Scientists, Syntheses and Investigation of Biologically Active Compounds, Riga (1989), p. 56.
M. E. Omar A.-Mohsen, N. S. Habib, and M. Adoulwafa Omaima, Pharmazie, 32, 758 (1977).
G. M. Badger, J. H. Seidler, and B. J. Thomson, J. Chem. Soc., No. 12, 3207 (1951).
C. B. Reese, J. Chem. Soc., No. 5, 895 (1958).
V. M. Potapov, Stereochemistry [in Russian], Khimiya, Moscow (1988), p. 309.
V. Petrov and W. Wragg, J. Chem. Soc., No. 10, 1410 (1947).
B. R. T. Keene and G. L. Turner, Tetrahedron, 27, 3405 (1971).
H. Seidl, R. Huisgen, and R. Grashey, Chem. Ber., 102, 926 (1969).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1361–1364, October, 1991.
Rights and permissions
About this article
Cite this article
Mikhailovskii, A.G., Bakrin, M.I. Synthesis and alkylation of amidines of the phenanthridine series. Chem Heterocycl Compd 27, 1091–1094 (1991). https://doi.org/10.1007/BF00486804
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00486804