Abstract
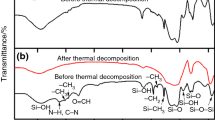
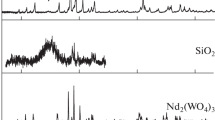
The thermal behaviour of samples with nominal composition 80/20=SiO2/SnO2, class B, 76.8/19.2/4.0=SiO2/SnO2/Sb2O5, class C1, and 76.8/19.2/2.0/2.0=SiO2/SnO2/Sb2O5/Sb2O3, class C2, is studied in the interval 25–1050°C by various instrumental methods. Results on these classes of samples, obtained from alkoxide precursors, are compared themselves and with samples of class A obtained from Si(OEt)4 and SnCl4. The segregation and crystallization of SnO2 occurs at 400°C in the presence of microdomains of SnO2·nH2O in the SiO2 gel matrix (class A), whereas it is observed at 700°C for samples B and C composed of Sn and Sb cations homogeneously dispersed in SiO2. This fact implies different mechanisms of SnO2 nucleation and growth. The crystallization of SiO2 is observed at 1200°C for samples A, at 1050°C for B and at 800°C for C. For this latter, the presence of Sb-oxide/ SiO2 reactive glass is invoked to the low-temperature crystallization of SiO2.
Similar content being viewed by others
Reference
G. Carturan, R. Ceccato, R. Campostrini, G. Principi, and U. Russo, J. Sol-Gel Sci. Technol., 5, 49 (1995).
G.W. Scherer, Yogyo-Kyokai-Shi, 95(1), 21 (1987).
R.C. Mehrotra, J. Non-Cryst. Solids, 121, 1 (1990).
H. Pentinghaus, J. Non-Cryst. Solids, 63, 193 (1984).
S. Sakka and K. Kamiya, J. Non-Cryst. Solids, 42, 403 (1980).
J.D. Mackenzie, J. Non-Cryst. Solids, 48, 1 (1982).
H. Dislich, J. Non-Cryst. Solids, 57, 371 (1983).
D.R. Uhlmann, M.C. Wienberg, and G. Teowee, J. Non-Cryst. Solids, 100, 154 (1988).
V.S. Nagarajan and K.J. Rao, J. Mater. Sci., 24, 2140 (1989).
W. Höland, E.R. Plumat, and P.H. Duvigneaud, J. Non-Cryst. Solids, 48, 205 (1982).
M.L. Balmer, F.F. Lange, and G.C. Levi, J. Amer. Ceram. Soc., 75, 946 (1992).
B.J.J. Zelinski, B.D. Fabes, and D.R. Uhlmann, J. Non-Cryst. Solids, 82, 307 (1986).
M. Nogami, J. Mater. Sci., 21, 3513 (1986).
J.C. Huling and G.L. Messing, J. Amer. Ceram. Soc., 74, 2374 (1991).
T. Ishikawa and S. Akagi, Phys. Chem. Glasses, 19(5), 108 (1978).
J.R. Taylor and A.C. Bull, in Ceramics Glaze Technology (Pergamon Press, Oxford, 1986) p. 139.
S. Enzo, S. Polizzi, and A. Benedetti, Z. Kristall., 170, 275 (1985).
S. Enzo, G. Fagherazzi, A. Benedetti, and S. Polizzi, J. Appl. Cryst., 21, 536 (1988).
R. Dal Maschio, S. Dirè, G. Carturan, S. Enzo, and L. Battezzati, J. Mater. Res., 7, 435 (1992).
J.C. Giuntini, W. Granier, J.V. Zanchetta, and A. Taha, J. Mater. Sci. Letters, 9, 1383 (1990).
J. Etchepare, M. Merian, and P. Kaplan, J. Chem. Phys., 68 (4), 1531 (1978).
M. Nogami, J. Non-Cryst. Solids, 69, 415 (1985).
M. Nogami and M. Tomozawa, J. Amer. Ceram. Soc., 69 (2), 99 (1986).
R. Dal Maschio, G.M. Del Felice, M. Filipponi, G.D. Sorarù, and G. Carturan, Am. Ceram. Soc. Bull, 71, 204 (1992).
L.M. Echeverria, J. Non-Cryst. Solids, 147 & 148, 559 (1992).
C.-C. Lin and P. Shen, J. Non-Cryst. Solids, 171, 281 (1994).
V. Jayram, C.G. Levi, T. Whitney, and R. Mehrabian, Mat. Sci. Eng., A124, 65 (1990).
D. Turnbull, J. Non-Cryst. Solids, 102, 117 (1988).
C.J. Brinker and G.W. Scherer, Sol-Gel Science. The Physics and Chemistry of Sol-Gel Processing (Academic Press, San Diego, 1990), p. 629.
R. Roy, J. Amer. Ceram. Soc., 52, 344 (1969).
V.J. Fratello, J.F. Hays, and D. Turnbull, J. Appl. Phys., 51, 4718 (1980);
V.J. Fratello, J.F. Hays, F. Spaepen, and D. Turnbull, J. Appl. Phys., 51, 6160 (1980).
M. Guglielmi and G. Principi, J. Non-Cryst. Solids, 48, 161 (1982).
C. Zhu, J. Phalippou, and J. Zarzycki, J. Non-Cryst. Solids, 82, 321 (1986).
F.A. Cotton and G. Wilkinson, in Advanced Inorganic Chemistry (J. Wiley & Sons, New York, 1980) p. 470.
R. Dal Maschio, S. Dirè, R. Campostrini, G.D. Sorarù, and G. Carturan, Mat. Res. Soc. Symp. Proc., 180, 351 (1990).
Author information
Authors and Affiliations
Additional information
G. Carturan, R. Ceccato, R. Campostrini, G. Principi, and U. Russo, submitted to J. Sol-Gel Sci. Tech.
Rights and permissions
About this article
Cite this article
Carturan, G., Ceccato, R., Campostrini, R. et al. SiO2/SnO2 and Sn/Sb-oxide/SiO2 gel-derived composites. Part 2: Thermal evolution and phase analysis. J Sol-Gel Sci Technol 5, 57–64 (1995). https://doi.org/10.1007/BF00486711
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00486711