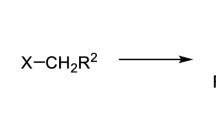
Abstract
Diazotization of 3-aminoisoxazolines has given stable, nonaromatic 2-isoxazoline-3-diazonium salts which undergo the azo-coupling reaction. The electronic structures of these diazo- and azo-compounds are discussed.
Similar content being viewed by others
Literature Cited
M. V. Gorelik, S. P. Titova, and V. I. Rybinov, Zh. Org. Khim., 16, 1322 (1980).
M. V. Gorelik, S. P. Titova, and V. I. Rybinov, Zh. Org. Khim., 21, 851 (1985).
V. I. Rybinov and M. V. Gorelik, Zh. Org. Khim., 23, 663 (1987).
L. A. Kazitsyna, B. S. Kikot', L. D. Ashkinadze, and O. A. Reutov, Dokl. Akad. Nauk SSSR, 151, 573 (1963).
A. T. Pilipenko, L. I. Savranskii, and V. L. Sheptun, Zh. Org. Khim., 24, 1746 (1988).
M. N. Glukhovtsev, B. Ya. Simkin, V. I. Minkin, M. V. Gorelik, V. I. Rybinov, and I. A. Yudilevich, Zh. Org. Khim. (in press).
W. Klötzer, H. Bretschneider, E. Fitz, R. Reiner, and G. Bader, Monatsh. Chem., 101, 1109 (1970).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 8, pp. 1110–1112, August, 1990.
Rights and permissions
About this article
Cite this article
Rybinov, V.I., Gorelik, M.V., Mustafina, M.Y. et al. Nonaromatic isoxazolinediazonium salts. Chem Heterocycl Compd 26, 929–931 (1990). https://doi.org/10.1007/BF00480873
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00480873