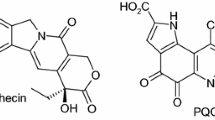
Abstract
The ethyl ester of Β-(2,3-dimethylindolyl-5)aminocrotonic acid under Vilsmeier reaction conditions is converted into isomeric linear and bent pyrroloquinolines with predominant formation of the latter. The enaminoketone which is obtained from the same aminoindole and dibenzoylmethane under the same conditions underoges the usual Comb cyclization with a change in the ratio for the yield of isomeric pyrroloquinolines.
Similar content being viewed by others
Literature Cited
S. A. Yamashkin, L. G. Yudin, and A. N. Kost, Khim. Geterotsikl. Soedin., No. 4, 493 (1983).
J. N. Dominquez, D. Adams, and J. A. Parez, Tetrahedron Lett., 24, 513 (1983).
A. N. Kost, S. A. Yamashkin, and L. G. Yudin, Khim. Geterotsikl. Soedin., No. 6, 770 (1977).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 2, pp. 228–230, February, 1989.
Rights and permissions
About this article
Cite this article
Yamashkin, S.A., Boriskina, N.Y. Synthesis of pyrroloquinolines. Chem Heterocycl Compd 25, 187–189 (1989). https://doi.org/10.1007/BF00479914
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00479914