Abstract
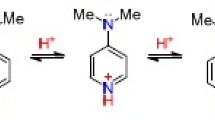
The dependence of the chemical shifts of the protons of quinoxaline and quinoxaline and pyrazine N-oxides and N,N-dioxides on the acid concentration was studied. The changes in the PMR spectra on passing from the neutral bases to the monocations were examined. The first protonation of pyrazine and quinoxaline N-oxides occurs at the N4 nitrogen atom.
Similar content being viewed by others
Literature cited
M. Kamiya, S. Katayama, and J. Akahori, Chem. Pharm. Bull., 17, 1821 (1969).
K. Tori, M. Ogata, and H. Kano, Chem. Pharm. Bull., 11, 235, 681 (1963).
D. J. Blears and S. S. Danyluk, Tetrahedron, 23, 2927 (1967).
R. A. Abramovitch and J. B. Davis, J. Chem. Soc., (B), 1137 (1966).
H. Kamei, J. Phys. Chem., 69, 2791 (1965).
P. J. Black and M. L. Heffernan, Austral. J. Chem., 17, 558 (1964).
E. Diaz and P. Joseph-Nathan, Spectrochim. Acta, A25, 1547 (1969).
A. H. Gawer and B. P. Dailey, J. Chem. Phys., 42, 2658 (1965).
V. M. S. Gil and J. N. Murrel, Trans. Faraday Soc., 60, 248 (1964).
M. M. Kaganskii, G. G. Dvoryantseva, and A. S. Elina, Dokl. Akad. Nauk SSSR, 197, 832 (1971).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 6, pp. 846–852, June, 1971.
Rights and permissions
About this article
Cite this article
Ul'yanova, T.N., Dvoryantseva, G.G., Alekseeva, L.M. et al. PMR spectra of protonated pyrazine and quinoxaline N-oxides. Chem Heterocycl Compd 7, 790–795 (1971). https://doi.org/10.1007/BF00476836
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00476836