Abstract
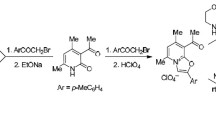
Stereoisomers of the corresponding 4-substituted 1,2,5-trimethyl-4-acetoxy- and 1,2,5-trimethyl-4-benzoxypiperidines were obtained by acylation of the geometrical isomers of 1,2, 5-trimethyl-4-piperidol and its 4-ethynyl, 4-ethyl, and 4-phenyl-substituted derivatives. The preferred conformations of the investigated esters were elucidated by means of the PMR and IR spectra.
Similar content being viewed by others
Literature cited
I. N. Nazarov, D. V. Sokolov, and V. N. Rakcheeva, Izv. Akad. Nauk SSSR, Otd. Khim. Nauk, 80 (1954).
I. N. Nazarov, V. Ya. Raigorodskaya, and V. A. Rudenko, Izv. Akad. Nauk SSSR, Otd. Khim. Nauk, 504 (1949).
I. N. Nazarov, I. A. Mokhir, B. V. Unkovskii, and G. S. Gusakova, Zh. Obshch. Khim., 29, 1867 (1959).
I. N. Nazarov, N. S. Prostakov, and N. I. Shvetsov, Zh. Obshch. Khim., 26, 2798 (1956).
V. I. Mamonov, G. G. Dvoryantseva, T. D. Sokolova, É. B. Sysoeva, and B. V. Unkovskii, Zh. Organ. Khim., 6, 1087 (1970).
T. F. Vlasova and Yu. N. Sheinker, Zh. Strukt. Khim., 11., 640 (1970).
I. N. Nazarov, B. V. Unkovskii, and I. A. Mokhir, Zh. Obshch. Khim., 30, 742 (1960).
I. N. Nazarov, N. S. Prostakov, N. N. Mikheeva, and O. A. Shavrygina, Zh. Obshch. Khim., 26, 2812 (1956).
N. N. Bolyard and S. M. McElvain, J. Am. Chem. Soc., 51, 922 (1929).
B. V. Unkovskii, I. A. Mokhir, and E. M. Urinovich, Zh. Obshch. Khim., 33, 1808 (1963).
C. I. Schmidle and R. C. Mansfield, J. Am. Chem. Soc., 77, 5698 (1955).
A. Ziering, L. Berger, S. D. Heineman, and J. Lee, J. Org. Chem., 12, 894 (1947).
D. Hirsch, in: Selected Problems of Stereochemistry [Russian translation], Mir, Moscow (1970), p. 199.
B. I. Ionin and B. A. Ershov, NMR Spectroscopy in Organic Chemistry [in Russian], Khimiya, Leningrad (1967), p. 221.
N. Bhacca and D. Williams, Applications of NMR Spectroscopy in Organic Chemistry [Russian translation], Mir, Moscow (1966), p. 69.
H. Rosenkrantz and S. Skrogström, J. Am. Chem. Soc., 77, 2837 (1955).
R. N. Jones and P. Herling, J. Am. Chem. Soc., 78, 1152 (1956),
O. V. Agashkin, G. S. Litvinenko, D. V. Sokolov, and S. S. Chasnikova, Zh. Obshch. Khim., 31, 862 (1961).
R. J. Ouellette, J. Am. Chem. Soc., 86, 3089 (1964).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No.1, pp. 50–55, January, 1973.
Rights and permissions
About this article
Cite this article
Sokolova, T.D., Kolosov, Y.V., Malina, Y.F. et al. Synthesis and three-dimensional structures of geometrical isomers of some 4-substitute d 1,2,5-trimethyl-4-acetoxy- and 1,2,5-trimethyl-4-benzoxypiperidines. Chem Heterocycl Compd 9, 42–46 (1973). https://doi.org/10.1007/BF00476147
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00476147