Abstract
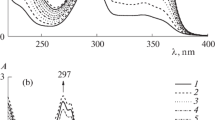
It is shown by means of IR and PMR spectra that the products of the aminolysis of esters of 3,3-dimethyl(2-carbethoxy)levulinic acid and γ-chloro-β,β,γ-trimethylbutyrolactone exist in the cyclic form of 2-hydroxy-5-pyrrolidones (Ia-d). It was established by PMR spectroscopy that, except for the 4-carbamido derivative (Id), these compounds undergo spontaneous dehydration in anhydrous solutions. In addition, ring-chain tautomeric equilibrium is established in solutions of the N-phenyl derivative (Ib). The rates of establishment of equilibrium and the equilibrium positions were investigated for both processes.
Similar content being viewed by others
Literature cited
B. M. Sheiman, L. Ya. Denisova, S. F. Dymova, I. M. Kustanovich, and V. M. Berezovskii, Khim. Geterotsikl. Soedin., 1190 (1971).
P. R. Jones, Chem. Rev., 63, 471 (1963).
W. Flitsh, Chem. Ber., 103, 3205 (1970).
R. Lukes and Z. Linhartova, Coll. Czech. Chem. Commun., 25, 502 (1960).
D. Hammond, in; Steric Effects in Organic Chemistry, M. S. Newman (editor), Wiley (1956).
B. V. Unkovskii, L. A. Ignatova, and M. G. Zaitseva, Khim. Geterotsikl. Soedin., 889 (1969).
I. Finkelstein, T. Willams, V. Toome, and S. Traiman, J. Org. Chem., 32, 3229 (1967).
B. M. Sheiman, L. Ya. Denisova, S. F. Dymova, I. M. Kustanovich, and V. M. Berezovskii, Zh. Organ. Khim., 5, 238 (1969).
Author information
Authors and Affiliations
Additional information
See [1] for communication VIII.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 1, pp. 22–26, January, 1973.
Rights and permissions
About this article
Cite this article
Sheiman, B.M., Denisova, L.Y., Dymova, S.F. et al. Syntheses of di- and tetrahydropyrroles. Chem Heterocycl Compd 9, 18–21 (1973). https://doi.org/10.1007/BF00476140
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00476140