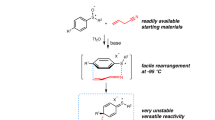
Abstract
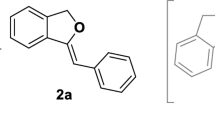
Treatment of allyl furfuryl ethers and sulfides with butyllithium results in metallation at the free α-position of the heterocycle, with partial Wittig rearrangement to the isomeric alcohols and sulfides and ring opening. With the benzo-derivatives, Wittig rearrangement and ring opening takes place.
Similar content being viewed by others
Literature Cited
J. E. Patrick and J. E. Baldwin, J. Am. Chem. Soc., 93, 3556 (1971).
J. Billman and I. Ducep, Tetrahedron, 36, 1249 (1980).
L. V. Mozhaeva, S. V. Shabanova, N. B. Kazennova, A. V. Anisimov, and E. A. Viktorova, Zh. Org. Khim., 20, 842 (1984).
C. W. Kamienski and D. H. Lewis, J. Org. Chem., 30, 3498 (1965).
I. A. Zoltewicz and G. G. Gaig, J. Am. Chem. Soc., 91, 550 (1969).
H. Steiberg, W. T. van Wijnen, and T. J. Boer, Tetrahedron, 28, 5423 (1972).
J. Z. Mortensen, B. Hedegaard, and S. O. Lawesson, Tetrahedron, 27, 3832 (1971).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 7, pp. 883–888, July, 1987.
Rights and permissions
About this article
Cite this article
Anisimov, A.V., Mozhaeva, L.V., Kazennova, N.B. et al. Wittig rearrangement of allyl furfuryl and allyl thienyl ethers and sulfides and their benzo-derivatives. Chem Heterocycl Compd 23, 724–729 (1987). https://doi.org/10.1007/BF00475634
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00475634