Abstract
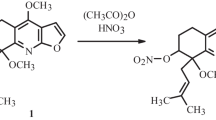
The direct nitration of brevicolline, brevicarine, and methyl-, acetyl-, and hydroxybrevicarine with concentrated nitric acid gave 6- and 8-nitrobrevicolline and 6,8-dinitro derivatives of the enumerated compounds. 6-Nitroharman was obtained by oxidation of 6-nitrobrevicolline to 6-nitroharman-4-carboxylic acid and subsequent decarboxylation.
Similar content being viewed by others
Literature cited
Brevicolline [in Russian], Kishinev (1969).
H. R. Snyder, J. Am. Chem. Soc., 70, 222 (1948).
I. P. Saxena, Indian J. Chem., 4, 148 (1966).
A. N. Lomakin and I. V. Terent'eva, Khim. Geterotsikl. Soedin., 1041 (1968).
J. Casini and L. Joodman, Can. J. Chem., 42, 1235 (1964).
L. B. Clapp and H. Lacey, J. Org. Chem., 33, 4262 (1968).
D. J. Rabiger and M. J. Chang, J. Heterocycl. Chem., 7, 307 (1970).
A. N. Lomakin, Zh. Vsesoyuzn. Khim. Obshchestva, 13, 354 (1968).
L. Bellamy, Infra-Red Spectra of Complex Molecules, Methuen (1958).
P. A. Vember, I. V. Terent'eva, and G. V. Lazur'evskii, Khim. Prirodn. Soedin., 249 (1967).
R. Bible, Guide to the NMR Empirical Method, Plenum (1967).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 7, pp. 987–990, July, 1972.
Rights and permissions
About this article
Cite this article
Shirshova, T.I., Terent'eva, I.V., Vember, P.A. et al. Nitration of alkaloids from carex parvae. Chem Heterocycl Compd 8, 898–901 (1972). https://doi.org/10.1007/BF00475229
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00475229