Abstract
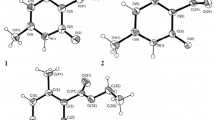
The interaction of 6-nitroazolo[1,5-a]pyrimidines with ethyl cyanoacetate is accompanied by transformation of the pyrimidine ring, forming derivatives of 2-azolylaminopyrimidine. The structure of the transformation products has been investigated in crystals and in solution.
Similar content being viewed by others
Literature Cited
V. L. Rusinov, A. A. Tumashov, T. L. Pilicheva, M. V. Kryakunov, and O. N. Chupakhin, Khim. Geterotsikl. Soedin., No. 6, 811 (1989).
V. L. Rusinov, T. L. Pilicheva, A. A. Tumashov, and O. N. Chupakhin, Khim. Geterotsikl. Soedin., No. 6, 857 (1987).
F. M. Allen, O. Kennard, D. C. Watson, L. Brammer, A. G. Orpen, and R. Tayler, J. Chem. Soc., Perkin Trans. 2, No. 1, 1 (1987).
L. J. Bellamy, Advances in Infrared Group Frequencies, Halsted Press, New York (1968).
V. L. Rusinov, I. Ya. Postovskii, A. Yu. Petrov, E. O. Sidorov, and Yu. A. Azev, Khim. Geterotsikl. Soedin., No. 11, 1554 (1981).
Author information
Authors and Affiliations
Additional information
For Communication 10, see [1].
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 12, pp. 1631–1637, December, 1990.
Rights and permissions
About this article
Cite this article
Rusinov, V.L., Pilicheva, T.L., Tumashov, A.A. et al. Nitroazines 11. Structure of products of transformation OF 6-nitroazolo-[1,5-a]pyrimidines under the influence of cyanoacetic ester. Chem Heterocycl Compd 26, 1357–1362 (1990). https://doi.org/10.1007/BF00473963
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00473963