Abstract
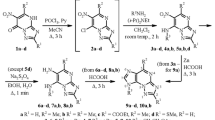
5-Diazoimidazole-4-carboxazide was isolated in the diazotization of 5(4)-aminoimidazole-4(5)-carboxhydrazide. The diazotization of N-substituted hydrazides of 5(4)-aminoimidazole-4(5)-carboxylic acis was studied. It is shown that the resulting diazo derivatives undergo cyclization to 3-arylideneaminoimidazo[4,5-d]-1,2,3-triazin-4-ones.
Similar content being viewed by others
Literature cited
V. A. Bakulev, V. S. Mokrushin, V. I. Ofitserov, Z. V. Pushkareva, and A. N. Grishakov, Khim. Geterotsikl. Soedin., No. 6, 836 (1979).
D. A. Peters and P. L. McGeer, Can. J. Physiol. Pharmacol., 46, 195 (1968).
D. H. R. Barton, R. E. O'Brien, and S. Sternhell, J. Chem. Soc., No. 2, 470 (1962).
Author information
Authors and Affiliations
Additional information
See [1] for communication 7.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 7, pp. 984–985, July, 1979.
Rights and permissions
About this article
Cite this article
Nifontov, V.I., Selezneva, I.S., Mokrushin, V.S. et al. Synthesis of analogs of 5(4)-aminoimidazole-4(5)-carboxamide and purines. Chem Heterocycl Compd 15, 805–806 (1979). https://doi.org/10.1007/BF00473569
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00473569