Abstract
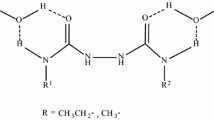
A new method of synthesis of the 1,3,2-dioxazolidine ring is developed based on the synthesis of 2-dimethylcarbamoyl-4,4-dimethyl-1,3,2-dioxazolidine by intramolecular cyclization of 1,1-dimethyl-3-(1,1-dimethyl-2-oxyethoxy)-3-chlorourea.
Similar content being viewed by others
Literature Cited
V. F. Rudchenko, I. I. Chervin, V. N. Voznesenskii, V. S. Nosova, and R. G. Kostyanovskii, Izv. Akad. Nauk SSSR, Ser. Khim., No. 12, 2788 (1988).
R. G. Kostyanovskii and G. V. Shustov, Dokl. Akad. Nauk SSSR, 232, 1081 (1977).
S. V. Varlamov, G. K. Kadorkina, and R. G. Kostyanovskii, Khim. Geterotsikl. Soedin., No. 3, 390 (1988).
V. F. Rudchenko, V. I. Shevchenko, and R. G. Kostyanovskii, Izv. Akad. Nauk SSSR, Ser. Khim., No. 7, 1556 (1987).
I. I. Chervin, V. S. Nosova, V. F. Rudchenko, V. I. Shevchenko, and R. G. Kostyanovskii, Khim. Geterotsikl. Soedin., No. 3, 396 (1988).
V. F. Rudchenko, V. I. Shevchenko, and R. G. Kostyanovskii, Izv. Akad. Nauk SSSR, Ser. Khim., No. 3, 598 (1986).
B. J. R. Nicolaus, G. Pagani, and E. Testa, Helv, Chim. Acta, 45, 358 (1962).
L. A. Simonyan, N. P. Gambaryan, P. V. Petrovskii, and I. L. Knunyants, Izv. Akad. Nauk SSSR, Ser. Khim., No. 2, 370 (1968).
J. Leitich, Angew. Chem. Int. Ed. Engl., 15, 372 (1976).
I. L. Charlton, C. C. Liao, and P. de Mayo, J. Am. Chem. Soc., 93, 2463 (1971).
V. F. Rudchenko, V. G. Shtamburg, A. P. Pleshkova, Sh. S. Nasibov, I. I. Chervin, and R. G. Kostyanovskii, Izv. Akad. Nauk SSSR, Ser. Khim., No. 11, 2539 (1981).
Author information
Authors and Affiliations
Additional information
Number 69 in the series “Asymmetric nitrogen,” for Number 68, see [1]; Number 43 in the series “Geminal systems,” for Number 42, see [1].
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 3, pp. 393–395, March, 1989.
Rights and permissions
About this article
Cite this article
Rudchenko, V.F., Shevchenko, V.I. & Kostyanovskii, R.G. New method of synthesis of the 1,3,2-dioxazolidine ring. Chem Heterocycl Compd 25, 330–332 (1989). https://doi.org/10.1007/BF00472396
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00472396