Abstract
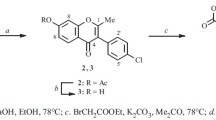
3-Hetaryl-7-methoxychromones were obtained by reaction of α-(5-ethoxycarbonyl-2-furyl)-, α-(2-benzofuryl)-, and α-(2-methoxy-carbonyl-5-benzofuryl)-2-hydroxy-4-methoxyacetophenones with methyl formate or ethyl orthoformate, and 3-hetaryl-4-hydroxy-7-methoxy-coumarins were obtained by reaction of the same compounds with diethyl carbonate. Methods for the synthesis of 3-hetaryl-7-hydroxy-chromones from α-furyl- and α-benzofuryl-substituted, 2,4-dihydroxyacetophenones were investigated.
Similar content being viewed by others
Literature cited
V. P. Khilya, L. G. Grishko, and V. Szabo, Khim. Getertotsikl. Soedin., 1317 (1972).
V. P. Khilya, L. G. Grishko, N. V. Sukharenko, and V. Szabo, Zh. Organ. Khim., 8, 1085 (1972).
V. P. Khilya, L. G. Grishko, L. L. Gontar′, and V. Szabo, Khim. Geterotsikl. Soedin., 1324 (1972).
V. P. Khilya, L. G. Grishko, L. I. Shevchenko, L. I. Barvinok, V. A. Dymovich, and V. Szabo, Khim. Geterotsikl. Soedin., 1202 (1973).
M. Yu. Kornilov, V. P. Khilya, and L. G. Grishko, Zh. Organ. Khim., 9, 2568 (1973).
H. S. Mahal, H. S. Rai, and K. Venkataraman, J. Chem. Soc., 1120 (1934).
M. L. Wolfrom and J. E. Mahan, J. Amer. Chem. Soc., 63, 1253 (1941).
V. R. Sethe and K. Venkataraman, Current Sci., 18, 373 (1949).
W. D. Ollis, in: The Chemistry of Flavonoid Compounds (edited by T. A. Geissman), Oxford-London — New York-Paris (1962), p. 385.
V. Prey, Ber., 74, 1219 (1941).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 2, pp. 174–179, February, 1975.
Rights and permissions
About this article
Cite this article
Szabo, V., Grishko, L.G., Borbei, S. et al. Chemistry of heteroanalogs of isoflavones. Chem Heterocycl Compd 11, 147–151 (1975). https://doi.org/10.1007/BF00471385
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00471385