Abstract
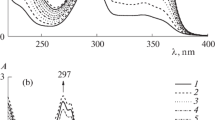
The polarographic behavior of a number of N-benzylideneiminopyridinium iodides in aqueous alcohol buffer solutions was studied over a wide range of pH values. The mechanism of their reduction on a dropping mercury electrode and the factors affecting the cleavage of the N-N bond are discussed. The reduction product-the pyridinium ion-induces the catalytic evolution of hydrogen.
Similar content being viewed by others
Literature cited
T. V. Troepol'skaya and Yu. P. Kitaev, Khim. Geterotsikl. Soedin., 1219 (1973).
P. Tompkins. Anal. Chem., 21, 64 (1949).
S. G. Mairanovskii. Dokl. Akad. Nauk SSSR. 110, 593 (1956).
S. G. Mairanovskii. Dokl. Akad. Nauk SSSR. 114, 1272 (1957).
L. Floch. M. S. Spritzer, and P. J. Elving, Anal. Chem., 38, 1074 (1966).
Yu. P. Kitaev and T. V. Troepol'skaya, Izv. Akad. Nauk SSSR, Ser. Khim., 1903 (1967).
Yu. P. Kitaev, I. M. Skrebkova, and L. I. Maslova, Izv. Akad. Nauk SSSR. Ser. Khim., 2194 (1970).
S. G. Mairanovskii, J. Electroanal. Chem., 12, 547 (1966).
Author information
Authors and Affiliations
Additional information
See [1] for communication XXXVIII.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 5, pp. 666–669, May, 1974.
Rights and permissions
About this article
Cite this article
Kitaev, Y.P., Troepol'skaya, T.V. & Orlova, L.N. Hydrazones. Chem Heterocycl Compd 10, 576–578 (1974). https://doi.org/10.1007/BF00471331
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00471331