Abstract
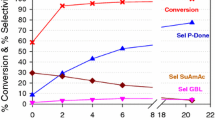
The influence of the nature of the solvent on the hydrogenation of succinimide over Raney nickel was studied. The greatest rates of reduction of the imide and of the formation of pyrrolidinone were found when alcohols and dioxane were used. An influence of the solvent on the ratio of the rates of the various stages in the synthesis of pyrrolidinone from the imide was found.
Similar content being viewed by others
References
I. Tafel and M. Stern, Ber., 33, 2225, 1900.
B. Sakurai, Bull. Chem. Soc. Japan, 10, 311, 1935.
A. Ladenburg, Ber., 20, 2215, 1887.
V. I. Romanovskii, A. I. Sokolova, and N. I. Tat'yanchikova, Khim. prom., 491, 1963.
US patent no. 3092638, 1963; RZhKh, 9N141P, 1965.
H. Adkins and H. Cramer, J. Am. Chem. Soc., 52, 4349, 1930.
L. M. Rice, E. E. Reid, and C. H. Grogan, J. Org. Chem., 19, 884, 1954.
K. C. Schreiber and V. P. Fernandez, J. Org. Chem., 26, 1744, 1961.
N. E. Vishnevskii, N. P. Glukhanov, and I. S. Kovalev, High-Pressure Apparatus with a Hermetic Drive [in Russian], Mashgiz, 1960.
D. V. Sokol'skii and F. Bizhanov, Vest. AN KazSSR, no. 6, 57, 1961.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Evgrashin, V.M., Ioffe, I.I. & Yakushkin, M.I. Hydrogenation of succinimide to pyrrolidin-2-one. Chem Heterocycl Compd 6, 330–333 (1970). https://doi.org/10.1007/BF00471235
Issue Date:
DOI: https://doi.org/10.1007/BF00471235