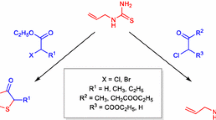
Abstract
Acrylonitrile reacts with dithiocarbaminates to give nitriles of N-alkyl-S-thiocarbaminylthiohydracrylic acids, which are then hydrolyzed to the corresponding amides and acids. Dehydration of the latter gives 3-alkyl-2-thiothiazon-4-ones. The UV spectra of N-alkyl-S-thiocarbaminylthiohydracrylic acids, and amides and nitriles of them are characterized by two maxima in the 245–250 nm (thioamide band) and 266–280 nm (dithiocarbonate band) regions. Thiazan ring closure results in bathochromic shift of absorption maxima due to lengthening of the conjugated chain. The average values of the atomic refraction for sulfur in the molecules of the compounds investigated correspond to a thioketone structure.
Similar content being viewed by others
References
J. Seyden-Penne, Ann. Chim., 3, 599, 1958.
J. L. Garraway, J. Chem. Soc., 4072, 1962.
E. V. Vladzimirskaya, Author's abstract, Thesis, L'vov, 1965.
E. Campaigne and P. K. Nargund, J. Med.Chem., 7, 132, 1964.
B. F. Goodrich, British Patent 626486, 1949.
J. E. Jansen, United States Patent 2709706, 1955.
A. E. Gillam and E. S. Stern, An Introduction to Electronic Absorption Spectroscopy in Organic Chemistry [Russian Translation], IL, Moscow, 1957.
S. S. Batsanov, Structural Refractometry [in Russian], Moscow, 1959.
E. V. Vladzimirskaya, Ukr. khim. zh., 30, 941, 1964.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Turkevich, N.M., Zimenkovskii, B.S. Some characteristic properties of 3-alkyl-2-thiothiazon-4-ones and of intermediates in their synthesis. Chem Heterocycl Compd 3, 667–671 (1967). https://doi.org/10.1007/BF00468341
Issue Date:
DOI: https://doi.org/10.1007/BF00468341