Abstract
Active transport of α-aminoisobutyric acid (AIB) in Vibrio costicola utilizes a system with affinity for glycine, alanine and, to some extent, methionine. AIB transport was more tolerant of high salt concentrations (3–4 M NaCl) in cells grown in the presence of 1.0 M NaCl than in those grown in the presence of 0.5 M NaCl. The former cells could also maintain much higher ATP contents than the latter in high salt concentrations.
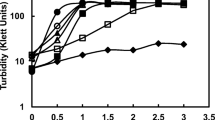
Transport kinetic studies performed with bacteria grown in 1.0 M NaCl revealed three effects of the Na+ ion: the first effect is to increase the apparent affinity (K t) of the transport system for AIB at Na+ concentrations <0.2 M, the second to increase the maximum velocity (V max) of transport (Na+ concentrations between 0.2 and 1.0 M), and the third to decrease the V max without affectig K t (Na+ concentrations >1.0 M). Cells grown in the presence of 0.5 M or 1.0 M NaCl had similar affinity for AIV. Thus, the differences in salt response of transport in these cells do not seem due to differences in AIB binding. Large, transport-inhibitory concentrations of NaCl resulted in efflux of AIB from cells preloaded in 0.5 M or 1.0 M NaCl, with most dramatic efflux occurring from the cells whose AIB transport was more salt-sensitive. Our results suggest that the degree to which high salt concentrations affect the transmembrane electrochemical energy source used for transport and ATP synthesis is an important determinant of salt tolerance.
Similar content being viewed by others
Abbreviations
- AIB:
-
α-aminoisobutyric acid
- pmf:
-
proton motive force
References
Bakker EP (1982) Membrane potential in a potassium transport-negative mutant of Escherichia coli K12. Biochim Biophys Acta 681:474–483
Booth IR, Kroll RG (1983) Regulation of cytoplasmic pH (pHi) in bacteria and its relationship to metabolism. Biochem Soc Trans 11:70–72
Fontana DR, Haug A (1982) Effects of sodium chloride on the plasma membrane of halotolerant Dunaliella primolecta: an electron spin resonance study. Arch Microbiol 131:184–190
Forsyth MP, Kushner DJ (1970) Nutrition and distribution of salt response in populations of moderately halophilic bacteria. Can J Microbiol 16:253–261
Galinsky EA, Trüper HG (1982) Betaine, a compatible solute in the extremely halophilic phototrophic bacteria Ectothiorhodospira halochloris. FEMS Microbiol Lett 13:357–360
Garcia ML, Guffanti AA, Krulwich TA (1983) Characterization of the Na+/H+ antiporter of alkalophilic bacilli in vivo: ΔΨ dependent 22Na+ efflux from whole cells. J Bacteriol 156: 1151–1157
Hamaide F, Kushner DJ, Sprott GD (1983) Proton motive force and Na+/H+ antiport in a moderate halophile. J Bacteriol 156:537–544
Hamaide F, Sprott GD, Kushner DJ (1984) Energetics of sodium-dependent α-aminoisobutyric acid transport in the moderate halophile Vibrio costicola. Biochim Biophys Acta 766:77–87
Hammes GG (1982) Enzyme catalysis and regulation. Academic Press, New York
Hanna K, Bengis-Garber C, Kushner DJ, Kogut M, Kates M (1984) The effect of salt concentration on the phospholipid and fatty acid composition of the moderate halophile, Vibrio costicola. Can J Microbiol 30:669–675
Henneman DH, Umbreit WW (1964) Influence of the physical state of the bacterial cell membrane upon the rate of respiration. J Bacteriol 87:1274–1280
Jarrell KF, Sprott GD (1983) Measurement and significance of the membrane potential in Methanobacterium bryantii. Biochim Biophys Acta 725:280–288
Knowles CJ, Smith L (1971) Effect of osmotic pressure of the medium on the volume of intact cells of Azotobacter vinelandii and on the rate of respiration. Biochim Biophys Acta 234:144–152
Kushner DJ, Hamaide F, MacLeod RA (1983) Development of salt-resistant active transport in a moderately halophilic bacterium. J Bacteriol 153:1163–1171
Lanyi JK (1974) Salt-dependent properties of proteins from extremely halophilic bacteria. Bacteriol Rev 38:279–290
Lanyi JK (1979) The role of Na+ in transport processes of bacterial membranes. Biochim Biophys Acta 559:377–397
LeRudulier D, Bouillard L (1983) Glycine betaine, an osmotic effector in Klebsiella pneumoniae and other members of the Enterobacteriaceae. Appl Environ Microbiol 46:152–159
Mitchell P (1966) Chemiosmotic coupling in oxidative and photosynthetic phosphorylation. Biol Rev Camb Philos Soc 41:445–502
Niven DF, MacLeod RA (1980) Sodium ion-substrate symport in a marine bacterium. J Bacteriol 142:603–607
Pettitt CA, Davidson VL, Cobb A, Knaff DB (1982) Sodium-dependent α-aminoisobutyrate transport by the photosynthetic purple sulfur bacterium Chromatium vinosum. Arch Biochem Biophys 216:306–313
Shkedy-Vinkler C, Avi-Dor Y (1975) Betaine-induced stimulation of respiration at high osmolarities in a halotolerant bacterium. Biochem J 150:219–226
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hamaide, F., Sprott, G.D. & Kushner, D.J. Energetic basis of development of salt-tolerant transport in a moderately halophilic bacterium, Vibrio costicola . Arch. Microbiol. 140, 231–235 (1984). https://doi.org/10.1007/BF00454933
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00454933